Drying is a demanding unit operation where solvents need to be removed without altering product chemistry. Vacuum dryers are essential pieces of equipment for accomplishing this task
Drying solids is a demanding task that requires both knowledge of process technology and a sound mechanical design of the equipment used. This article provides an overview of batch-drying principles and possible options for vertical vacuum-contact dryers.
Most solids in the chemical process industries (CPI) are recovered by precipitation or crystallization. The suspended solids are mechanically separated by filtering or centrifuging in the first instance. In most solid-liquid-separation processes, thermal evaporation does not compare favorably with mechanical separation by filtration or centrifugation with respect to energy efficiency. However, when it comes to the final moisture content, thermal evaporation is often the follow-up step to mechanical dewatering methods in order to achieve the desired dryness.
The residual moisture content of the solids after filtration or centrifugation is influenced by the properties of the solid’s crystal structure. A typical filtration cake can show residual moisture content of between 5 and 45%. To dry a wet cake to lower values, thermal evaporation is an effective way to achieve a true “dry” solids powder.
For some products, continuous drying processes, such as belt and conveyor drying or spray drying, are suitable methods to achieve a dry powder in a single pass. While continuous systems are not a subject of this article, more information can be found in Part 2 of this Feature Report, “Optimizing Analysis for Spray Drying Processes”.
Vacuum drying
During any drying process, all additives and solvents have to be removed from the solid material without changing its original chemical composition. In essence, there must be no changes to the molecular structure during the drying process and no derivatives formed due to the drying process.
A vacuum contact dryer is built to evaporate solvents from a solid by the use of the following physical and mechanical parameters: temperature; low pressure; and mechanical fluidization by agitation.
In most cases, vacuum contact dryers are restricted by the requirement that they not influence the chemistry of the product. However, special processes are possible, where the application of high temperature will initiate or control a chemical reaction.
The degree of residual moisture in the finished product is strictly related to the use of the product and its specifications. Examples include the following: less than 5% moisture for waste material for disposal; less than 3% in foodstuffs for storage considerations; less than 1% in herbal extracts; and less than 0.1% in active pharmaceutical ingredients (APIs).
A vacuum contact dryer can achieve these values for the separation of solids from solvents and there are cases where the solids, the solvents or both, are valuable products.
Batch drying process
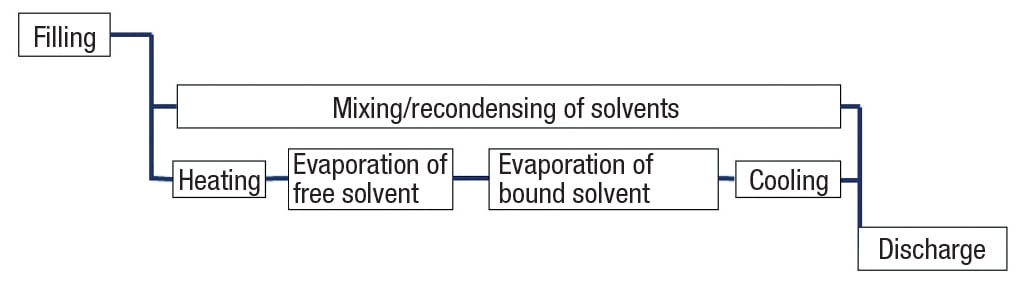
Almost all batch drying processes consist of the process steps mentioned in Figure 1. Whether the steps are performed in a non-agitated tray or in an agitated vessel, the concepts discussed here for evaporation and re-condensing and collecting the solvents are the same.
Each process step in batch drying has individual issues that require special attention:
Filling.During filling, factors such as the use of gravity or force (pumping), as well as the need for a contamination-free transfer into the dryer will have to be addressed.
Mixing. In order to create an even temperature and solvent-concentration distribution in the bulk material, as well as to support the outflow, mixing is mandatory in the dryer. However, the influence of shear forces created by the mixing action on the solids needs to be addressed.
Re-condensing. Whether the purpose of the drying is to retain the dry power and discard the solvent, or vice-versa, is determined by the user. Re-condensing and collection of the solvents is a demanding task in the process.
Evaporation of free solvent. The solvent selection dictates the evaporation conditions (enthalpy) and the maximum temperature limits of the solids dictates the thermal limitations for the drying system.
Evaporation of bound solvent. Some solvents tend to stick to the solid particles by physical bridging, or to hide in the pores of the particle structure, while others don’t. In order to predict required process times, detailed knowledge of the separation behavior is critical.
Cooling. For safe discharge, it might be required to cool the solids to very low temperatures prior to discharge. Since the heat removal from a dry power is subject to even lower heat-transfer coefficients, the cooling step might be time consuming.
Discharge. While fluids or suspensions drain well from a vessel, draining a dry powder can be a challenge. Residues after draining are lost in most instances and reduce the cost efficiency of the dryer.
In addition to the aforementioned basic requirements, supplemental codes and standards, such local laws, pressure vessel regulations, good manufacturing practice (GMP) rules, pharmaceutical standards, U.S. Occupational Safety and Health Administration (OSHA) requirements and explosion protection measures have to be considered.
Equipment overview
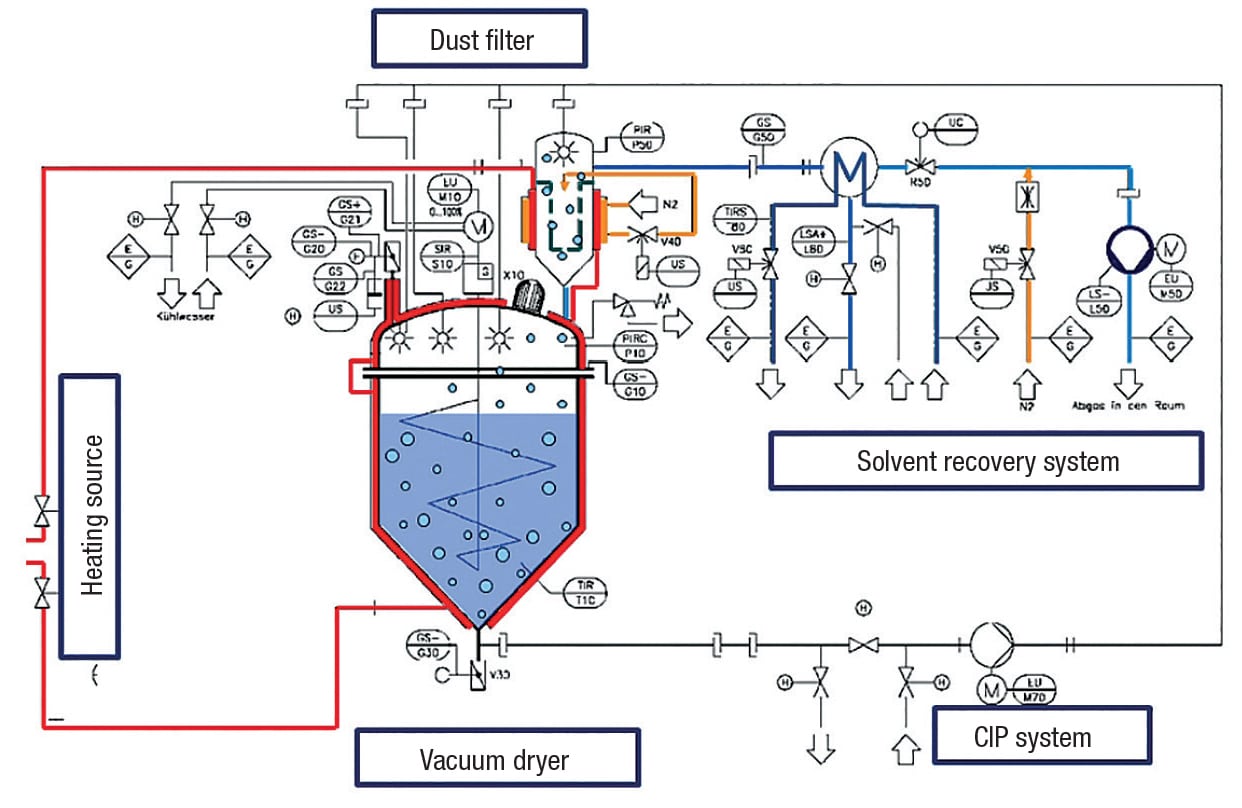
The following section is intended to identify the key components of a vacuum drying system (Figure 2).
Heat source. With respect to the heating source for the dryer, available site utilities, as well as product specifications have to be considered. While a supplier of aluminum pigments might ban water as a heating fluid for fire hazard reasons, an API producer might avoid thermal oil to avoid contamination risks. Both are secondary aspects, not directly related to the physical operation of heating.
The base specification for a dryer heating system should consider the following:
- Provide a sufficient amount of heat (Joules) in an energy efficient way
- Provide measures to keep a constant temperature in the system
- The correct heating power and capacity calculation has to consider the following:
- Initial heating of all wetted components (equipment) within a reasonable amount of time
- Provide sufficient energy to heat the product load to the operating temperature
- Provide sufficient energy to evaporate the solvent load at the given conditions
- Compensate for energy losses
- Maintain temperature
In most cases, circulating a clear fluid in a closed loop is the appropriate method for transporting the energy from the heat source to the dryer vessel. For thermal fluids, water, water-glycol mixes or thermal oils are common.
For its high specific-heat capacity, water is the first choice as a thermal fluid when the temperature needs to stay below 150°C. At process temperatures above 150°C, thermal oil becomes more feasible, since oil shows only a marginal pressure rise at elevated temperatures.
Whether the closed loop is driven by one heat exchanger for heating and one heat exchanger for cooling, or the system is fed by a centralized plant heating and cooling system is to be decided based on the site conditions. Proper sizing is important.
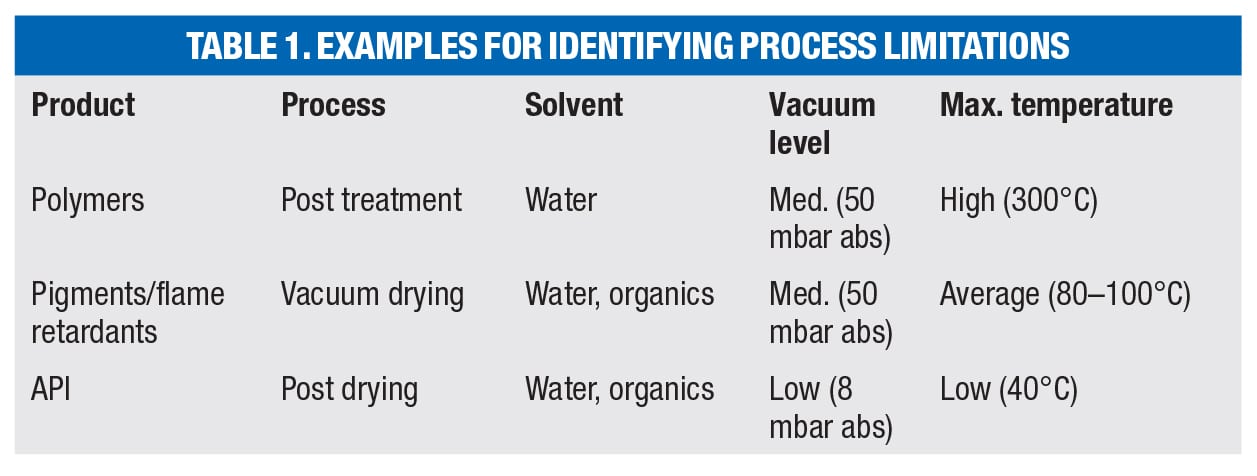
Prior to any equipment selection, the physical limitations for the process need to be defined by the process owner. Each and every process has special demands, including (but not limited to) the three examples in Table 1.
Vacuum dryer design
The majority of commercially available vacuum contact dryers are built according to the same principle: a jacketed vessel — either horizontal or vertical — with a wall-wiping agitator. Vertically oriented dryers can take advantage of gravity to influence product flow (Figure 3). Vertical vacuum-contact dryers include the following:
- An upright cylindrical vessel with a filling-to-height diameter ratio designed for bulk mixing
- A cone bottom to support the outflow of solids
- A jacketed to suit the selected heating system
- A top-entry agitator, designed for solids applications (re-starting in settled solids)
- Valves and safety applications to operate a pressure vessel
Solids impellers circulate the entire contents of the vessel from the gravity filling until the gravity discharge. The impellers can be hydrofoil type, designed to create maximum flow with the lowest possible power input.
Energy balance is important. Mixing power by the impeller equals the power into the process. In a temperature-sensitive process, excessive mixing power input might disturb the fragile temperature balance in the product.
The key feature of all agitators in a vacuum contact dryer is an agitator system that operates with minimum wall clearances and wipes the full wetted surface of the vessel. Wall clearances from 5 to 15 mm are common and feasible, and require both a precisely fabricated vessel and a deflection-free balanced impeller.
Since the process is typically operated under vacuum, the vessel must be fabricated according to the American Society of Mechanical Engineers (ASME) Boiler and Pressure Vessel Code (BPVC), and must also follow local laws and regulations, as applicable.
Dust filter
By heating a wet solid in the closed vessel, evaporation will start. The solvents will be condensed in an external solvent-recovery system. This migration of solvents will create a strong gas flow that may cause dust formation as the solids dry.
Commercially available solvent-recovery systems are built for heavy duty services, but not for high amounts of dust. In order to prevent the plugging of lines — or simply to keep the solids where they belong, in the dryer vessel — a dust filter is installed on top of the dryer vessel.
Modern dryers feature heated filter domes with automatic blowback cleaning systems that are properly designed to protect the solvent-recovery system from the dust.
Two types of commercially available filter elements are discussed here, but others’ experience might include many more systems.
The dust-loaded area of the dryer is separated from the solvent recovery line by a gas-tight filter plate. This filter plate carries the filter elements. One possibility is a fabric bag filter mounted on a wire cage, while the other is a sintered metal or wire mesh (10µm) cartridge made of metal wire or metal fibers
The selection of one of these filter systems is based on owner preference and on the overall operating conditions (including temperature). While the fabric bag filters are disposable and low in replacement cost, the metal cartridge is re-useable with a high lifetime expectation.
In both cases, proper sizing of the filter is critical. An undersized filter will lead to an excess pressure drop in the solvent-recovery line, with effects on the total evaporation regime.
Solvent-recovery system
As the vacuum contact dryer evaporates the solvents, the solvent-recovery system needs to re-condense the solvent, usually at the same rate. There are many ways to achieve this objective. This article covers one method that has shown some advantages when it comes to collecting the solvent for further use (Figure 4).
A robust vacuum pump (liquid-ring pump or dry-running pump) creates the initial low pressure that will activate the process. Design criteria for the pump includes the following:
- Evacuate the total system volume in a reasonable time
- Maintain the vacuum level under changing process conditions
- Compensate for air leakage across the full system
- Compensate for inert gas volumes introduced during the process
In essence, the vacuum pump has to keep the system running while also covering for losses.
The key component of the solvent-recovery system is the heat exchanger or condenser that is installed on the low-vacuum side of the pump. Properly designed and activated with the proper cooling fluid, solvents evaporated in the hot dryer vessel will migrate to the cold surface of the heat exchanger and will be re-condensed.
Condensation on the vacuum side of the pump will result in a huge draft — the condenser acts as a “vacuum pump.” The liquefied solvents are collected in a collection vessel for further use or transfer. Cooling or insulating the vessel is recommended when the system is installed at higher ambient temperatures.
With respect to the cooling methods, again the circulation of a clear fluid is one of the first choices. Based on commonly used solvents and the vacuum level achieved, anything from water (at 8°C) to brine (at –15°C) might be required. One item needs to be considered: water at 25°C should not be considered “cold.”
Collecting the solvents is the most precise method for back-calculating the residual moisture in the dry product. This can be done simply by measuring the solvent volume. A small amount of solvents, however, might escape even a properly designed condenser. To catch the saturated solvents, a second condenser in the exhaust line of the vacuum pump might be required.
Process controls
All of the components mentioned in this article require safe and precise operation. The large number of signals to be processed and valves to be handled does not allow manual operation of such a system, except situations involving small-scale tests in a laboratory environment. Automatic process control systems that focus on a proper temperature and vacuum regime are mandatory for industrial vacuum drying.
Drying process steps
As noted earlier in the article, an effective drying process requires knowledge of the full picture of the process, including filling, mixing, re-condensing, evaporation of free solvent, evaporation of bound solvent, cooling and discharge of products
Filling. There are many ways of transferring a solvent-loaded wet cake into a dryer vessel. This could be the subject of an article on its own, covering all aspects such as contamination, operator exposure levels, explosion risks and dosing the correct amount. Some dryers provide a wide opening at the top of the dryer vessel where valves, dosing or conveying systems, including (but not limited to) material-containment apparatus, can be installed. Gravity will support the filling process.
Mixing. During the filling process, the mixer may or may not run. However, it has to be designed for re-starting in settled solids at any time during the process. Stalling during a drying process should be avoided. In some rare cases, however, stopping the agitator is even required to overcome a certain process condition. In essence, an “off-the-shelf” agitator will not do the job. Dryer agitators have to be designed for the purpose.
The degree of mixing might or might not affect the drying time. It is the task of the scale-up engineer to predict the influence of the mixing on the drying behavior. Engineers must gain this information in a small-scale test.
Re-condensing. Evaporation starts when the solvent vapor pressure exceeds the pressure in the vessel or the product temperature exceeds the boiling temperature of the solvent.
Since the heat transfer into a mixed solids bed is difficult (because of low overall heat-transfer coefficients of about 50 W/m²K), additional measures are required to increase the system efficiency. Often the dry product shows temperature sensitivity, limiting the allowable heating fluid temperature.
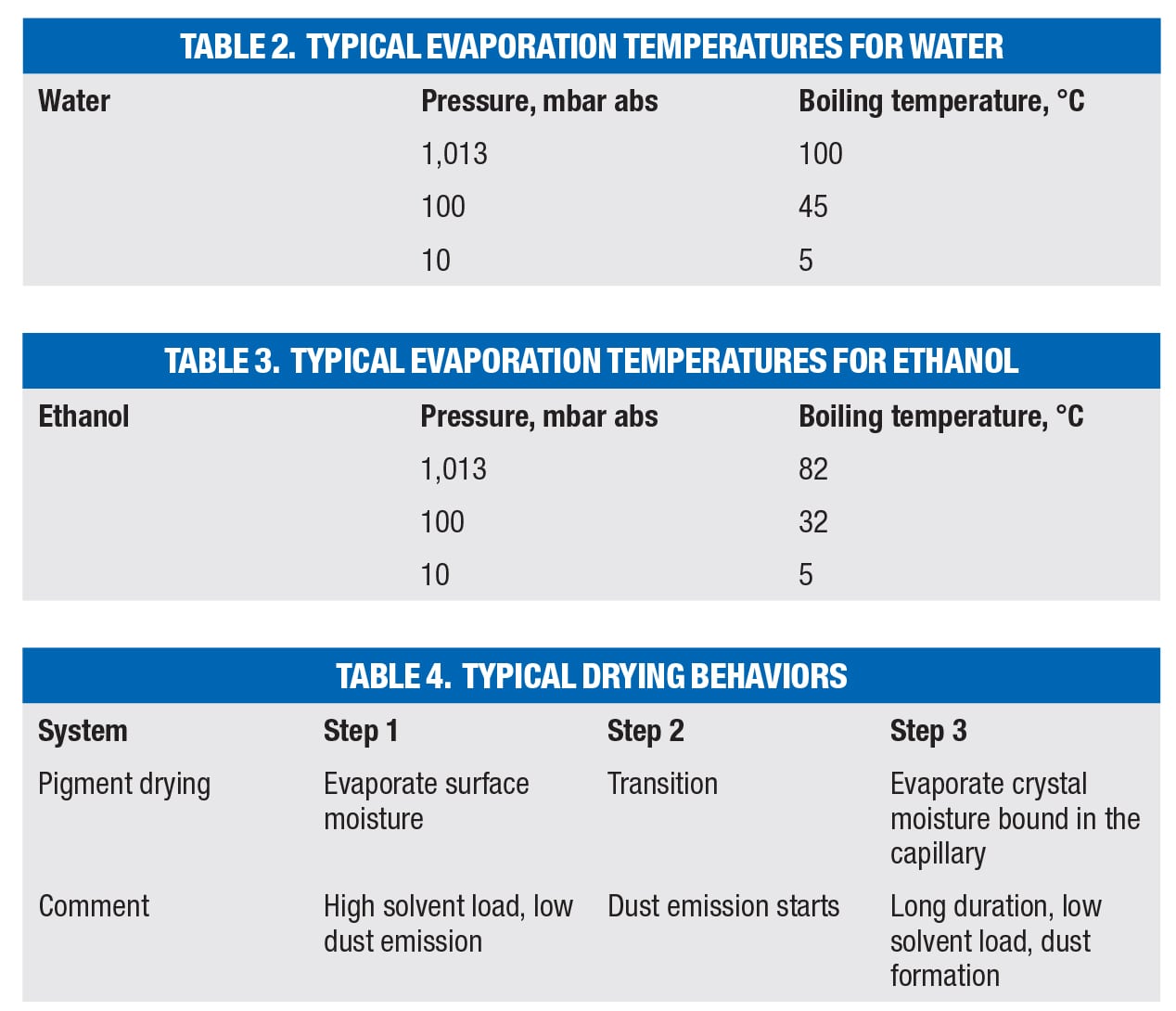
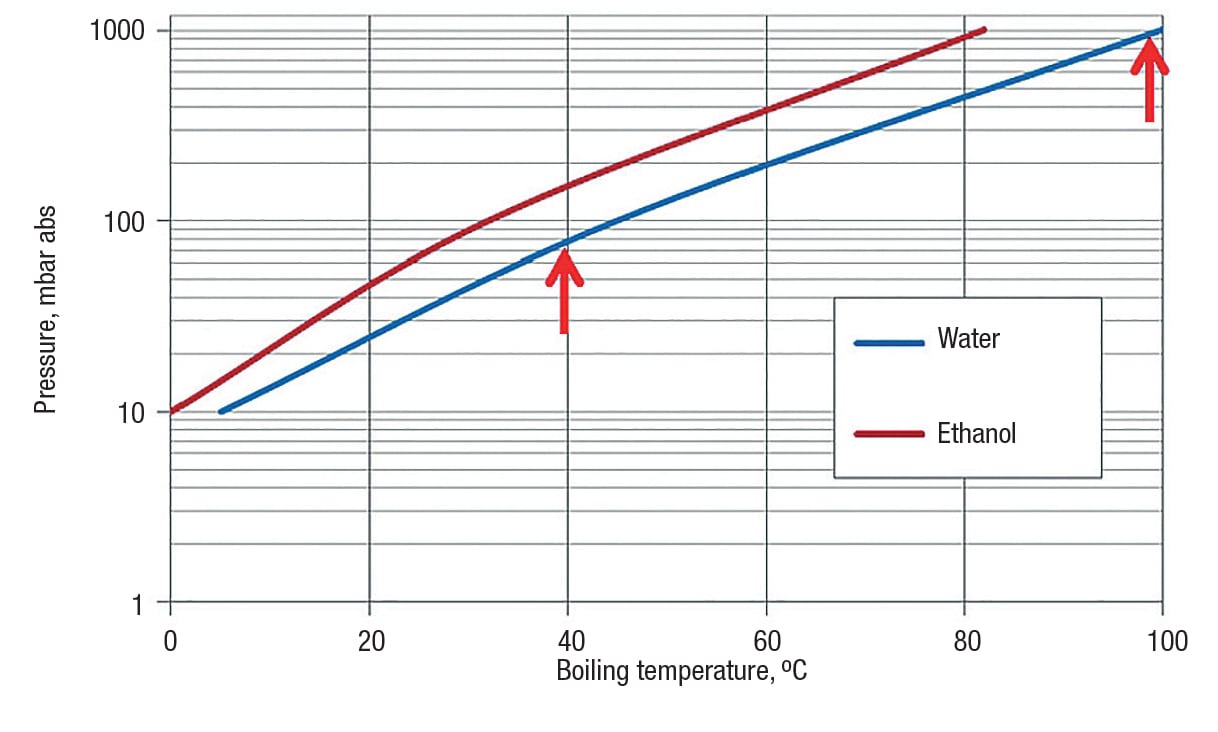
In order to compensate for both, the limitations in heat transfer and heating temperature, working under vacuum is advised. By lowering the pressure in the vessel, the boiling temperature of solvents drops. Typical evaporation temperatures for water and ethanol are shown in Tables 2 and 3.
Water has a vapor pressure of 1,013 mbar abs (~760 torr) at 100°C, so its vapor pressure would exceed atmospheric pressure at temperatures higher than 100°C and the water starts boiling (Figure 5).
If a desired solid product is water-loaded, but melts above 40°C, it is advisable to ensure that the heating-system temperature does not exceed 40°C. The vapor pressure of water at 39°C is 70 mbar abs. To avoid melting during the drying process, the pressure inside the vessel has to be set to less than 70 mbar abs.
Drying curve. Once the theoretical parameters have been set, a small-scale test is recommended to gain sufficient information about the exact drying behavior of the product. The following are characteristics for typical small-scale testing of drying behavior:
- 3-L scale: Yes/No-type testing with limited data collection for scaleup
- 25-L scale: best for free-flowing products; results allow safe scaleup to plant size
- 50–100-L test: best for difficult-flowing products; results allow safe scaleup to production scale
During a drying test, in the first instance, proof that the agitator is suitable to move the product is mandatory. All process-related parameters must be collected in order to generate the drying chart, a plot that shows the progression of the solvent content over the drying time (Figure 6).

Figure 6. Drying curves show the progression of solvent content in the solid material over the drying time
For this example process, the solvent evaporates at an almost constant pace until reaching the goal of 5% solvent by weight. Based on the results of this chart, the following parameters affecting the hardware are calculated:
- Required power consumption of the heating system (amount of evaporation heat in relation to the time required)
- Required dust filter sizing, with respect to the solvent load over the evaporation time
- Required vacuum condenser sizing, with respect to the solvent load over the evaporation time
Each and every drying chart looks different for the great number of drying applications. However, they all give an indication about typical drying behaviors and are divided into three sections (Table 4).
Discharge. When the process temperature is equal to the heating temperature, this is an indication that energy is no longer being spent for evaporation, and the drying process can stop. However, safety considerations might not allow the discharge of a hot solid. In many cases, cooling prior to discharge to lower temperatures is mandatory. Some dryer systems have the ability to change the heating system to a cooling mode. While the heat transfer for heating already has very low values, the values for cooling a dry powder are even lower. Cooling the dry solids will consume additional production time. In addition, the stainless-steel mass of the dryer equipment also needs to be cooled. In cases where time is of the essence, it might be beneficial to separate the functions and transfer the hot solids by closed pipe into a designated cooling vessel directly below the dryer. This procedure saves the energy required to cool the equipment mass.
Maximum yield after discharge is important for the cost balance of the drying system. In a single-product manufacturing process, leaving residues in the drying vessel for the next batch may be permitted. However, in a typical pharmaceutical or fine chemicals process, “cross-contamination” of products is banned.
With a cleverly designed vessel and a flexible agitator, the equipment can be made to suit the flow characteristics of the product. A vertical vessel will, by nature, drain better than a horizontal drying vessel. A cone bottom supports outflow far better than a flat or dished bottom. The agitator must be in a position to rotate at elevated speeds, including the changing the direction of rotation for discharge. Also, surface polishing and surface linings can help to reduce friction as the solid product flows.
Scaleup. Scaling up a drying process requires both small-scale testing capabilities and scaleup experience. Once successfully dried at a 25-, 50- or 100-L scale, scaleup rules can be used to predict the following:
- Drying time
- Solvent load, energy demand
- Residues after discharge
- Particle size changes due to shear on the product
- Wear of the agitator parts due to exposure to the product
As a basic rule, a safe scaleup requires that the geometry of the small-scale test dryer is geometrically similar to the production-size dryer.
Scaleup works best when the trials have been carried out under identical physical parameters (such as pressure and temperature) as the full-scale process. If this is not possible, correction factors have to be applied to the scaleup formula. The scaleup rules consider the following items:
- The ratio of volume-to-heated-surface-area of the small-scale dryer to the full-scale dryer
- A temperature correction factor in moving from small scale to full scale, if the values deviate from one another
- A factor to account for the influence of agitation speed on the evaporation
If all data are available, safe scaleup by a factor of 200 is readily achievable.
Concluding remarks
The information included here describes the basic concept of a vacuum contact-drying system. This type of system can be applied to many chemical applications by designing the components accordingly. The following are some of the typical situations:
- Selection of wetted parts with respect to corrosion (most commercially available metals)
- Designing the components for GMP standards (food and pharmaceutical industries)
- Designing the components for critical solvents, such as organic solvents
- Designing the components for severe site conditions (outdoor and weather protected)
- Designing the components for high demands on cleanability (cleanroom environment)
- Designing liquid cleaning systems (clean-in-place; CIP) for the dryer vessel
- Designing the components for out-of-specification applications (high temperatures and pressures)
Aside from plant experience and knowledge of the process, cooperation with a capable dryer company opens a wide range of possibilities beyond vacuum contact drying. Applications include solvent extraction from herbs and spices; all-in-one reaction systems for paints and pigments; food pasteurization and drying; and all-in-one chemical reactor/dryers.
Edited by Scott Jenkins
Author
 Eberhard Tritschler is product manager for sales at Ekato Systems GmbH (Käppelemattweg 2, 79650 Schopfheim, Germany; Phone: +49 7622-6907-808; Email: [email protected]; Website: www.ekato.com). Tritschler has served in sales and application engineering at Ekato Systems since 2005, and prior to that, worked in sales and application engineering at several other Ekato divisions. Tritschler holds a bachelor’s degree in mechanical engineering from the Technical School of Engineering in Lörrach Germany, and has also earned a degree in business administration from the HWK School in Schopfheim, Germany. Aside from his day-to-day business duties in the sales organization of Ekato Systems, Tritschler also speaks at customer seminars, tradeshows and business schools. He has special expertise in the mechanical aspects of vacuum contact drying, as well as extractions and all-in-one reactions in batch-type mixing vessels.
Eberhard Tritschler is product manager for sales at Ekato Systems GmbH (Käppelemattweg 2, 79650 Schopfheim, Germany; Phone: +49 7622-6907-808; Email: [email protected]; Website: www.ekato.com). Tritschler has served in sales and application engineering at Ekato Systems since 2005, and prior to that, worked in sales and application engineering at several other Ekato divisions. Tritschler holds a bachelor’s degree in mechanical engineering from the Technical School of Engineering in Lörrach Germany, and has also earned a degree in business administration from the HWK School in Schopfheim, Germany. Aside from his day-to-day business duties in the sales organization of Ekato Systems, Tritschler also speaks at customer seminars, tradeshows and business schools. He has special expertise in the mechanical aspects of vacuum contact drying, as well as extractions and all-in-one reactions in batch-type mixing vessels.