Catalysis is among the most important chemical phenomena in industrial chemistry, as well as in many biological and research fields. This one-page reference provides a review of several of the fundamental concepts underlying catalysis. 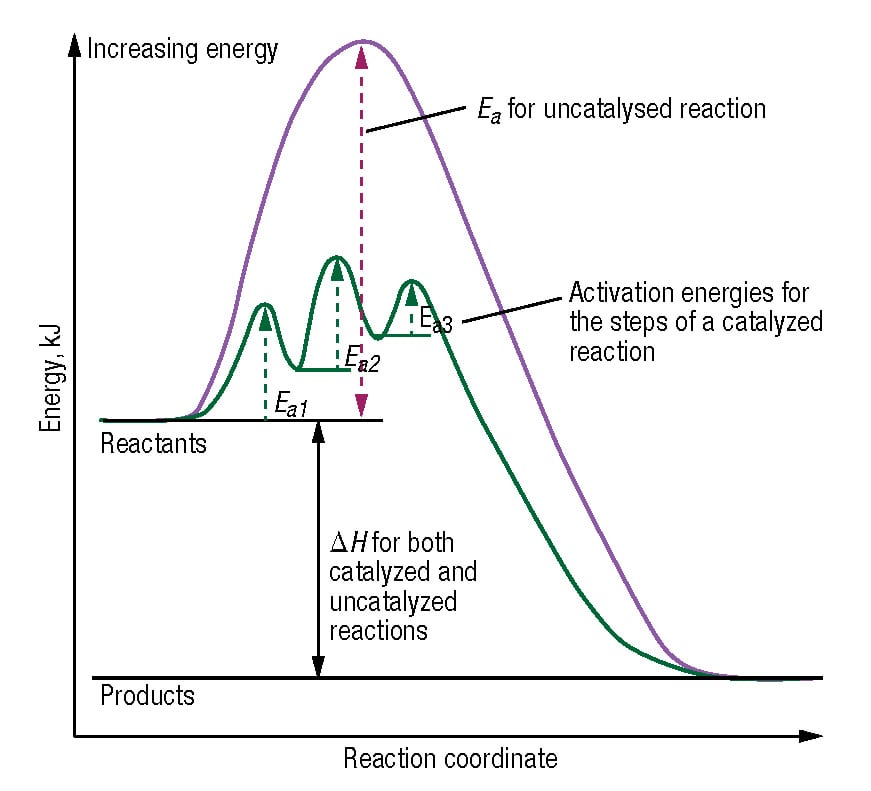
Catalyst basics
A catalyst interacts with chemical reactants to increase the reaction rate. Catalysts form fleeting intermediate chemical complexes with reactants, allowing the reaction to follow a different mechanistic pathway that requires lower activation energy (Ea) than the corresponding uncatalyzed reaction. Ea is often thought of as an energy barrier over which the reactants must pass to form products. Activation energies are often shown on graphs that plot reaction coordinate against thermodynamic free energy (Figure). Reaction coordinates are one-dimensional representations of the progress of a chemical reaction. Catalysts are broadly categorized as homogeneous or heterogeneous. Homogeneous refers to those catalysts that are dissolved in the reaction medium, forming a single phase with the reactants. Heterogeneous catalysts exist as a distinct phase from the reaction mixture and are often porous solid particles. Both categories are important for industrial chemistry. Examples of liquid-phase, acid-base-catalyzed reactions include hydrolysis of esters and amides, enolization of aldehydes and ketones, esterification of alcohols, halogenation of acetone and others. Heterogeneous catalysts play a key role in the production of petrochemicals, including cracking, alkylation, polymerization, isomerization, dehydrogenation and many others.
Mechanism of action
Most chemical reactions involve simultaneous (rather than sequential) bond breaking and bond forming. Along the pathway of reactants to products, the molecules adopt a configuration that represents the highest potential energy state, known as the transition state. The transition state is characterized by bonds that are both partially formed and partially broken. Catalysts form an intermediate species with one of the reactants and stabilize the transition state, allowing the reaction to proceed with a mechanism that requires lower energy. As products form, the catalyst is regenerated.
Catalyst features
The following summarizes key catalyst characteristics: Reversible reactions. In the case of reversible reactions, the catalyst acts on both the forward and reverse reactions. The catalyst does not affect the position of the equilibrium, but it does accelerate the rate at which equilibrium is reached. Energy. The presence of catalyst does not affect the potential energy of the reactants or products. It affects only the activation energy. Selectivity. In reactions with multiple feasible mechanisms, catalysts can often exhibit selectivity by binding to the transition state in a way that favors one reaction pathway over others. Catalyzed reactions often show a different product distribution than the same uncatalyzed reaction. Reaction rate. In homogeneous-catalyzed reactions, the reaction rate is generally proportional to the concentration of the catalyst, while in heterogeneous catalysis, the reaction rate is proportional to the surface area of the solid catalyst and the concentration of active centers (catalytic sites).
Solid-catalyzed reaction steps
Solid-catalyzed, fluid-phase chemical reactions generally undergo the following steps: Diffusion. Reactants in the fluid phase diffuse to the exterior surface of a catalyst particle and into the catalyst pores Adsorption. Reactants adsorb to the active centers in catalyst pores Reaction. The surface-adsorbed reactants form products Desorption. The product molecules desorb from the exterior surface of the catalyst pores Diffusion. Product molecules diffuse back into the bulk fluid In most cases, one of these steps contributes most significantly to the overall reaction rate, and often the others steps can be ignored or combined when determining reaction rates. The significance of each step depends heavily on the reactants and the reaction conditions.
Influencing factors
The following are factors that can play a large role in determining which step is more or less significant:
- Fluid-dynamic factors
- Catalyst properties (such as particle size, porosity, pore geometry and surface characteristics)
- Diffusion characteristics of fluid reactants and products
- Activation energy requirements for adsorption and desorption of reactants and products to and from solid surfaces
- Overall Ea of the catalyzed reaction
- Thermal factors (temperature and heat-transport characteristics)
References
1) Perry, R.H. and Green, D.W., “Perry’s Chemical Engineering Handbook,” 7th ed., McGraw Hill Professional, Section 4, Chapter 12. 1997.
2) Wijngaarden, R.J. and others, “Industrial Catalysis: Optimizing Catalysts and Processes,” Wiley-VCH, Weinheim, Germany, 1999.
3) University of Texas, Chemistry 302. Course material on chemical kinetics. Accessed from ch302.cm.utexas.edu, April 2015.
Department Editor: Scott Jenkins