Lignin bio-oil is a common waste stream formed during the production of cellulose and ethanol from biomass. Due to its high oxygen and low hydrogen content, lignin bio-oil must be upgraded via hydrogenation and deoxygenation before it can be used as a fuel. Furthermore, because of the very low hydrogen solubility in water and high activation energy of deoxygenation, lignin bio-oil upgrading typically requires processing at very high temperatures and hydrogen pressures.
Now, a team of scientists from Georgia Institute of Technology (Atlanta, Ga.; www.gatech.edu), led by professor Yulin Deng, has studied a solution-based, dual-catalyst system for combined hydrogenation and deoxygenation of lignin bio-oil that operates at ambient conditions (diagram). They found that polyoxometalate acid (SiW12) can react with H2, transfer H+ to water, and release hydrogen as an active species inside the water in the presence of a platinum-based catalyst.
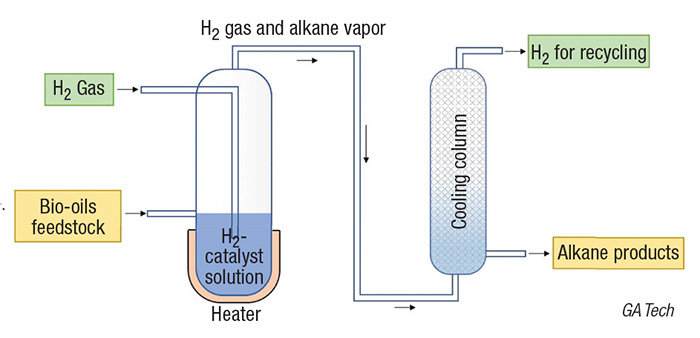
The team also observed that SiW12 can significantly reduce the activation energy of deoxygenation. As a result, SiW12 can act as both a hydrogen-transfer agent and a deoxygenation catalyst, which overcomes issues associated with hydrogen’s low solubility in water at low pressures and the high activation energy of deoxygenation.
So far, the technology has been demonstrated in a laboratory-scale batch reactor. According to Deng, the process is versatile enough to produce many products beyond fuels, including benzene, cyclohexane, toluene, cyclohexanol and many others. “The key parameters to control the product selectivity include the type of catalyst, reaction time and temperature. Using different catalysts, the same lignin monomer may be converted to different final products,” adds Deng. “We have only tested platinum and palladium so far, and we found the ratio of the final products shifts, which means the product selectivity can be adjusted by using different catalysts. This is one of our future research topics.” The team is also looking at how lignin monomers can be partially hydrogenated or hydrodeoxygenated in the early stages of the reaction, and how the reaction progression over time can be controlled to yield different final products.