Monoethylene glycol (MEG), also known as ethylene glycol (EG) or simply glycol, is a diol mostly used for the production of polyester fibers and polyethylene terephthalate (PET) resins. It is also used in antifreeze applications and in pharmaceuticals and cosmetics. MEG is conventionally produced through the hydrolysis of ethylene oxide (EO), which itself is obtained via ethylene oxidation.
The process
Figure 1 depicts MEG production from ethylene via a process similar to the OMEGA catalytic process from Shell Global Solutions (The Hague, the Netherlands; www.shell.com). In the described process, MEG is produced via EO, which is manufactured in an integrated plant utilizing Shell EO technology. An important feature of the process is the negligible production of diethylene glycol (DEG) and triethylene glycol (TEG), which occur as byproducts in other ethylene glycol production processes.
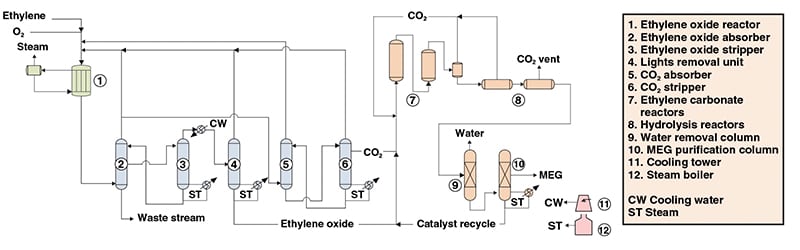
Figure 1. Monoethylene glycol (MEG) production, according to a process similiar to the Shell OMEGA process
Ethylene oxide production. Ethylene and oxygen are fed to a multi-tubular reactor, forming EO. This exothermic reaction, conducted in fixed beds in the reactor tubes, occurs in the gaseous phase with the use of a silver catalyst supported on alumina. Steam is generated by the heat of reaction.
Ethylene oxide recovery. The reactor product stream is fed to the EO absorber for lights removal by water quenching. Part of this gaseous overhead stream is recycled to the reactor, while the other part is sent to a carbon-dioxide-removal unit composed of an absorber and a stripper. In this unit, CO2 is separated to be used in ethylene carbonate production.
A diluted EO stream removed from the absorber is fed to the EO stripper, where it is concentrated and recovered in the overheads. The crude EO stream is condensed. Residual light gases are recovered from it and recycled to the reactor. The resulting EO stream is directed to the next section.
Ethylene glycol production and purification. Ethylene oxide is reacted with CO2, forming ethylene carbonate, which is then hydrolyzed to form MEG and CO2. Both reactions are carried out in the liquid phase using homogeneous catalysts.
CO2 streams from the reaction steps are recycled to the ethylene carbonate reactor. MEG is purified in two distillation columns where water is removed, leading to the final MEG product. The catalyst is separated and recycled to the ethylene carbonate reactors.
Economic performance
An economic evaluation of the process was conducted based on data from the first quarter of 2015, assuming a facility with a nominal capacity of 750,000 ton/yr of MEG constructed on the U.S. Gulf Coast.
Estimated capital expenses (total fixed investment, working capital and initial expenses) to construct the plant are about $630 million, while the operating expenses are estimated at about $620/ton of MEG.
Global perspective
Shell OMEGA is the first process to enable ethylene glycol production via a fully catalytic process. According to the licenser, the process is able to achieve EO-to-EG converstion and selectivity near 100%, leading to production of MEG only.
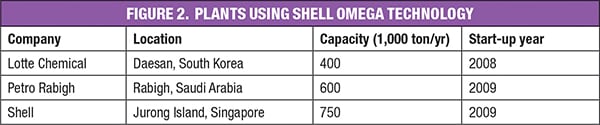
However, although 40% of worldwide ethylene glycol production is derived from processes using Shell technologies, only three plants in the world use the new Shell OMEGA technology (Figure 2). As can be seen, two of these plants are located in Asia, which is the region of the world responsible by the major share of MEG global consumption. China alone accounts for about 45% of global demand for MEG.
Edited by Scott Jenkins
Editor’s Note: The content for this column is supplied by Intratec Solutions LLC (Houston; www.intratec.us) and edited by Chemical Engineering. The analyses and models presented are prepared on the basis of publicly available and non-confidential information. The content represents the opinions of Intratec only. More information about the methodology for preparing analysis can be found, along with terms of use, at www.intratec.us/che.