Small-scale distillation processes have specific concerns that are different than commercial-scale distillation. Presented here are several topics, including column internals and column-sizing software, considered from the perspective of pilot-scale distillation
The design and operation of laboratory- and pilot-scale columns are greatly influenced by their purpose, the effect of scale on engineering principles, the availability of equipment, and the short-term nature of projects. Some of the main considerations are listed here, and these often require innovative solutions to meet the project requirements:
• There are significant differences in the type of equipment available, such as pumps, flowmeters, and control values, the types of trays and packing used, and the facilities required. The facilities can also affect the column design, especially regarding column height
• For separations problems in which data are the main product, small-scale equipment must have appropriate sample points or on-line analyses capabilities, and instrumentation that may not be necessary for an equivalent commercial column, and often must be able to operate at a wider range of conditions than for a commercial column
• For each separation, there are always many options, and it is usually a compromise based on equipment size, type, and features, which are influenced by cost, time, and equipment and facilities availability
• The short-term nature of most laboratory- and pilot-scale projects emphasize the importance of equipment versatility and ease of assembly. Flexible systems that can be easily modified are very desirable
In a previously published article (Chem. Eng., March 2023, pp. 23–30), we highlighted the specific concerns imposed on the purpose, design, and operation of small-scale distillation equipment and discussed how these concerns manifest themselves in the actual equipment that is typically used. The topics included the challenges of scale, high-vacuum operation, heat control, reflux control, distillate and sidestream product takeoffs, and reboilers.
This article continues the discussion by covering additional aspects that should be considered when dealing with small-scale distillation equipment. The following topics are discussed:
• Column internals
• Flexible distillation column concept
• Column-sizing software
• Decanters
• Free-radical polymer formation
Column internals
Column internals, whether trays, structured packing or random packing, play a large role in distillation effectiveness. In pilot-scale processes, there are a number of design considerations relative to column internals, and some key differences compared to large-scale distillation.
Trays. Glass Oldershaw-style trays [1] are commonly used for laboratory- and small pilot-scale distillation columns. Figure 1 shows an example of an unjacketed section. They are available as “off-the-shelf” components in a range of diameters from 28 to 100 mm, with up to 30 trays per section (usually in five-tray increments), in vacuum-jacketed or bare column sections. The trays available from each vendor have dimensions for the number of holes, hole diameter, and downcomers that are fairly similar. Custom variations of the column sections for themowells, feed or sample ports and connection joints are common. Custom designs for the tray dimensions are possible, but tend to have high setup costs.

FIGURE 1. Glass Oldershaw trays are available “off the shelf”
Due to the high degree of mixing for liquid on the trays and for the vapor, the efficiency for Oldershaw trays is a point efficiency [2]. For commercial trays, in which the liquid on the entire tray is not as well mixed, the composition can vary significantly as the liquid moves across the tray, which increases the overall tray efficiency above the point efficiency. The extreme case for this is a Lewis Type 2 efficiency, where the vapor is not mixed and the liquid flow is entirely in one direction. Large-column efficiencies can exceed 100%, depending on the definition of efficiency [3]. Therefore, if a specific separation can be achieved in the laboratory with a certain number of Oldershaw trays, the separation in a commercial column would normally be expected to be at least as good with the same number of trays. This makes it relatively easy to scale up Oldershaw column data. One caveat is that this conclusion is not valid if cellular foam is present. Trays also have an advantage with high-purity service, since they ensure good mixing and their design prohibits channeling of gas and liquid, in which a small amount can lead to product impurities.
One of the other key advantages of using glass trays is the ability to observe the tray activity and determine if there are issues related to variation in tray activity between trays, foaming, fouling and color formation. The pressure drop is usually in the range of 1 to 2 mm Hg per tray. This is an advantage for laboratory columns with many trays or those that are operating under high vacuum, since the reboiler temperature will be not be excessively elevated due to high pressure drop in the column. However, care must be taken when scaling up these data, since commercial column trays will usually have significantly higher pressure drops. This means that not only will the reboiler temperature be higher for the same head pressure as the laboratory column, but the separation may actually be affected if there is a significant effect of pressure on the vapor-liquid equilibrium, which is especially true at vacuum conditions.
One of the disadvantages of using glass is the pressure constraint, which limits operation to atmospheric pressure (or very slightly above) and below. Metal Oldershaw column sections are not commercially available. It is possible to custom build these, but this option is difficult and expensive by traditional methods. However, work by the authors’ company, along with the Robert C. Byrd Institute, suggests that metal trays can be fabricated using additive manufacturing (3-D printing). Examples are shown in Figure 2. Each tray and its surrounding column are printed with appropriate grooves that easily allow several tray units to be welded together to create sections with any desired number of trays, as well as several different materials of construction, such as stainless steel, high-nickel alloys and titanium. Thermowells, sample ports and feed inputs can easily be added.

FIGURE 2. Oldershaw trays can also be made from 3D-printed stainless steel, as well as a variety of other metals
High precision is possible for the creation of these trays, including the hole dimensions and locations. They are relatively inexpensive to make and design customization is not difficult, which is an additional advantage over glass trays.
Random packing. This type of vapor-liquid contactor is often used for small-scale distillation columns as an alternative to trays. This consists of an open tube with a packing support, into which the packing can be poured. The HETP (height equivalent to a theoretical plate) is very low for most laboratory random packings, allowing for a relatively large number of stages in a location with limited height, such as under a typical laboratory fume hood. Pressure drop per stage is generally lower than for trays, making random packing especially good for vacuum operation. If fouling occurs in the packing, the packing can be easily replaced.
However, scaleup of distillation setups using random packing is more difficult than for Oldershaw trays. Liquid distribution and collection are important considerations, as well as the surface area of the column wall. For Oldershaw trays, the liquid from the tray above, or from a liquid feed stream, can simply be fed onto the tray where it will be mixed by the bubbling action on the tray. Packing requires that the liquid be properly distributed onto the packing. For columns up to about 3 in. in diameter, only a single tube located in the center of the column and just above the packing is sufficient for good liquid distribution (multiple points are required for larger-diameter columns). Between 2 and 3 in. in diameter, some additional packing may be required due to suboptimal distribution. This is not difficult for the introduction of reflux at the top of the column, but for feed sections, the liquid from the packed section above must be collected, mixed with the feed, then distributed to the packing section below.
It is also important that the random packing elements are not too large relative to the column diameter. The general rule-of-thumb is that the column diameter should be at least eight times the approximate diameter of the packing. However, it is important to check with packing suppliers for their recommendations.
Figure 3 shows examples of two common types of random packing. Figure 3 (left) shows ceramic saddles. These are very inexpensive and are resistant to acidic corrosion. However, they have relatively high pressure and high HETP. In addition, the smallest ceramic saddles are around 0.25 in. in diameter, which means that they should not be used in columns less than 2 in. in diameter.

FIGURE 3. Ceramic saddles (left; photo used by permission from Chemglass Life Sciences) and metal Pro-Pak packing (right; photo used by permission from Performance Distillation Solutions) are examples of random packing
Figure 3 (right) also shows an example of Pro-Pak [4] (Performance Distillation Solutions; spun off from Cannon Instrument Co.) packing. This packing has low pressure drop and low HETP and comes in two sizes (0.24 and 0.16 in.). It can be obtained in a variety of metals, including Hastelloy C. Distillation data are available to assist with column design. This type of packing is relatively expensive, but the cost is partially offset by the large number of stages per volume of packing purchased, for a given column diameter. At low vacuum, there is some uncertainty in efficiency, so HETP testing should be performed for a specific service.
Structured packing. Figure 4 shows an example of structured packing, which is available for laboratory- and pilot-scale columns from Koch-Glitsch [5] and Sulzer [6], such as DX and EX. This type of packing has the lowest pressure drop per theoretical stage of any vapor-liquid contactor discussed so far. Its most important application is in high-vacuum service, where minimizing pressure drop is important. The HETP can be relatively low also. Sulzer does not have scale up information for DX/EX gauze packing, only for CY and BX. Testing with model systems at the same pressure and flow rates can be used to compare the packing efficiency of DX/EX with commercial packing. At elevated pressure, the HETP for gauze packing can appreciably increase (more height required) as the liquid layer thickness builds on the gauze, eliminating the advantage of gauze over sheet metal.

FIGURE 4. Structured packing has a low pressure drop (photo courtesy of Sulzer Chemtech)
Structured packing has similar liquid distribution and collection requirements to those of random packing. Structured packing generally has long lead times (6 months) since it has to be custom-made for a specific column size. There are significant pricing variations depending on vendor/materials.
Vigreux column. This type of column, an example of which is shown in Figure 5, is often used by chemists to make crude separations. It has very low pressure drop and low liquid holdup, and is very inexpensive. For a 1-in. diameter column, the HETP is in the range of 4 to 6 in. [7]. Vigreux columns work well for small-scale separations where low pressure drop is needed (high-vacuum operation), a low number of stages is required, visual observation is desirable (Vigreux columns are constructed of glass), and there is a strong desire to minimize expenses.

FIGURE 5. Vigreux columns, such as the one shown here, are used for crude separations
Flexible distillation column
Flexible distillation columns refer to those designed to be reconfigurable for multiple options. This type of laboratory-scale column has reconfigurable reboilers and column sections.
Reboiler. Figure 6 shows a diagram of a flexible reboiler to be used as part of a versatile laboratory distillation apparatus. The glass vessel can be heated by hot oil in its jacket or its coil. A bottom port allows connection to a forced-circulation system. The glass vessel can be replaced by an unjacketed version that would allow for heat input by an electric heating mantle.

FIGURE 6. Flexible reboilers, like the one shown here, have a heating coil, jacket and metal headplate (drawing used with permission from H.S. Martin, part of the AGI Group)
The hollow headplate, which is constructed of Hastelloy C, can be heated with hot oil to prevent vapor condensation on the headplate. A magnetic-drive agitator is mounted in the center of the headplate, which also has connections for the column, a thermocouple, liquid level and column differential pressure (high lead), forced circulation return and sample collection. Agitation becomes critical at low pressure, due to liquid bumping (vapor-liquid disengagement).
Seals between the headplate and column, as well as the headplate and glass vessel, are made of FFKM perfluoroelastomeric O-rings (Kalrez, Simrez, Markez and so on), which are resistant to a large range of chemicals. Some formulations can withstand temperatures in excess of 300˚C, and provide nearly leak-free connections even at high vacuum. At elevated pressures, however, other joint types should be explored.
A reboiler such as this would be intended as the centerpiece of a versatile distillation system that will allow for rapid exchange of packed or trayed sections, as well as modifications to the number of stages, number and location of feeds, condenser types and inclusion (or not) of a vacuum system, a sidestream or an overhead decanter. The heating bath has the capability of heating oil up to 300˚C.
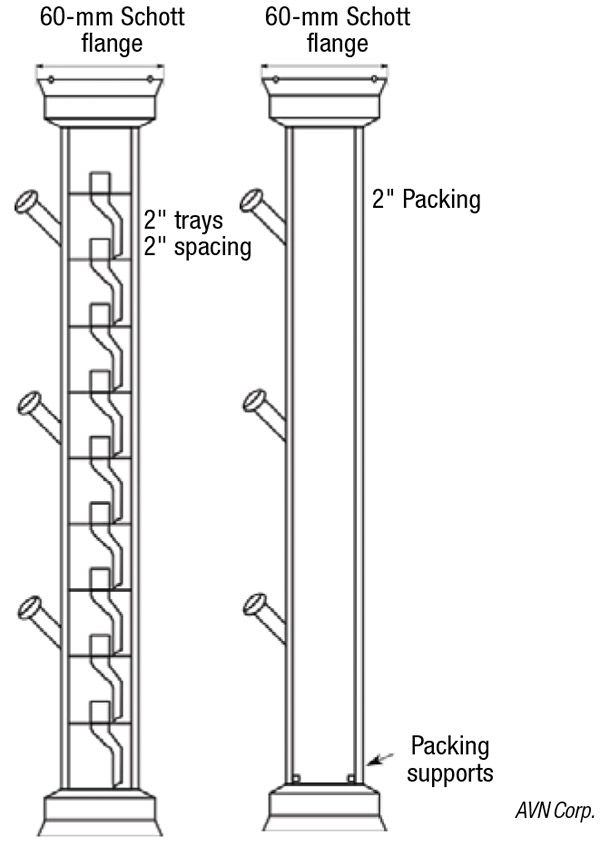
FIGURE 7. Flexible column sections can contain trays or packing
Column sections. Figure 7 shows examples of column sections to be used in this distillation system. These contain Oldershaw trays, or are open tubes used for random or structured packing. O-ring joints (60-mm Schott Duran O-ring joints) allow connection to the metal headplate discussed above, or to each other. Multiple O-ring ball-joint column ports can be used for feed, sampling or thermocouples. These types of joints have the advantage (over ground-glass joints) of low clearances, so that the entire column does not need to be disassembled to make a change to a column section. They do not require vacuum grease, which can lead to process contamination. The joints don’t freeze together, which would prevent disassembly, and they provide more consistent vacuum sealing performance. In addition, the clamps are easy to install. In order to prevent heat loss from the joints, custom joint heaters have been fabricated (Figure 8).

FIGURE 8. The photo shows a joint heater for 60-mm Schott flange on a 1-in.-dia. packed and unsilvered distillation column
Normally, adiabatic operation will be accomplished by heat tape and insulation. However, vacuum-jacketed column sections can also be used.
The 3D-printed tray sections, as discussed earlier, can be used in place of the glass sections for pressure operations, assuming that the other glass parts on the system will be replaced with metal. Alternative joint types will need to be used, such as sanitary seals, pipe, Grayloc and so on.
Column sizing software
Process-simulation software, such as Aspen Plus, is capable of determining the size requirement of trayed or packed commercial-scale columns. However, the type of trays and packing typically used in small-scale columns are not included in the column internals calculation blocks of the process simulation software.
Proprietary software for sizing laboratory- and pilot-scale trayed and packed distillation columns is available. This software allows for rapid comparisons between trayed and packed column to assist in the most appropriate type and size for column specifications. The programs are being converted to web-based tools. Other tools for small-scale distillation, such as for reboiler and condenser design, are anticipated in the future.
Decanters
For chemical systems with heterogeneous azeotropes, in which the distillate consists of two liquid phases, a decanter will usually be required. Two of the main concerns are (1) the residence time required for phase separation and (2) how the removal of each phase will be controlled. Determination of the residence time will not be covered here.
Depending on the ratio of the phases, the flowrates and the degree of sophistication of the equipment setup, decantation can sometimes be controlled manually. However, it is usually desirable for this operation to be automated.
The most simplistic means of automating a decanter is through the use of hydrostatic pressure [8]. Figure 9 shows an example of a pilot-scale decanter based on this principle. A dip tube enters the top of the decanter and extends to the approximate level of the liquid-liquid interface (the diptube generally has a momentum breaker). The light phase (usually organic) simply overflows from a port toward the top of the decanter, low enough to leave a vapor space above the liquid for venting. The heavy phase flows out the bottom of the decanter and into the bottom of an inverted U-tube, which is vented at the top of the U-tube. The U-tube has two joints that allow it to be rotated to adjust the height of the top of the U-tube relative to the decanter overflow drain. The hydrostatic head in the decanter, which consists of the light and heavy phase, must equal the head in the U-tube, which contains only the heavy phase. The height of the interface will automatically adjust itself until the hydrostatic pressures are equal. The interface would normally be maintained at a level in which the mass ratio of the two phases is equal to their ratio entering the decanter, so that the residence time of each phase is about the same.
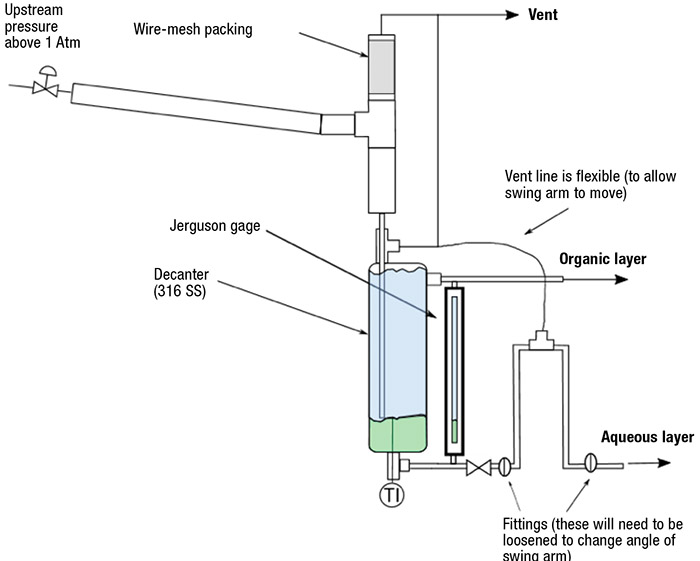
FIGURE 9. Automatic decanters can be used for distillates with two liquid phases
For the metal decanter depicted in Figure 9, a Jerguson gage is connected between the liquid overflow drain and the decanter drain to allow visual observation of the interface level inside of the decanter. In this design, the feed to the atmospheric decanter was vented from a higher pressure through a depressurization system to eliminate disturbances of the interface in the decanter.
Glass versions of this decanter, based on the same hydrostatic concepts, are routinely used for glass laboratory- and small-pilot-scale columns. For atmospheric columns, smooth operation of the decanter is usually easy to achieve. However, if the column operates under vacuum, or at elevated pressure (in a metal column), care must be taken to ensure that the decanter pressure is controlled well. Fluctuations in the pressure can cause a false pressure difference between the decanter and the U-tube, causing the interface to move. In these situations, as an alternative to the hydrostatic head principle, a capacitance probe or guided-wave radar, tied to a control valve or pump below the decanter bottom drain, can be used to control the interface level.
Figure 10 shows another style of decanter that uses the input from a guided-wave radar instrument to maintain the overall and liquid-liquid interface levels by controlling the flowrates of the upper and lower liquid layers. This type of instrument is able to detect both the overall and liquid-liquid interface levels because, as the radar signal is transmitted down the metal probe, part of the signal bounces from the top surface, but the remainder of the signal continues down to the liquid-liquid interface, where it is reflected back. The rod can be cut with a metal saw and can be replaced to change the length or materials of construction (possibly to an exotic metal, such as Hastelloy).
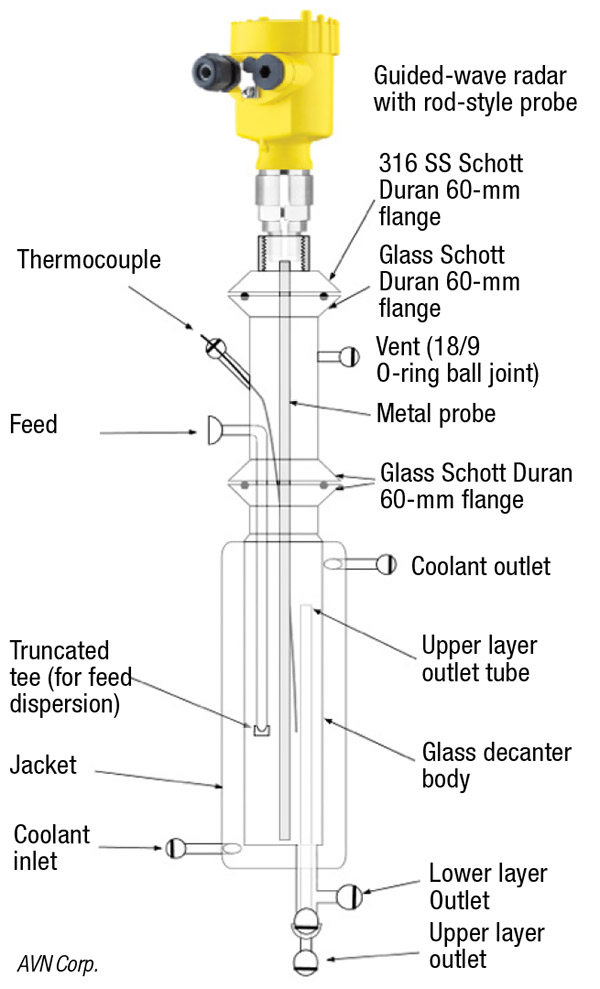
FIGURE 10. This diagram shows an automatic decanter that is controlled based on signals from a guided-wave radar instrument
The body of the decanter is glass (although it can be easily swapped out for an all-metal design), which allows for visual observation of both liquid layers. The decanter also has a jacket for coolant. The connection joints are O-ring ball joints, which allow for very good sealing without the use of grease. A small diameter (1/16-in.) thermocouple can be inserted down into the liquid. Feed material flows into the center of the decanter through a truncated tee that directs motion horizontally to minimize disturbances of the liquid-liquid interface.
The upper layer leaves the decanter through the upper-layer outlet tube. The lower layer flows out the bottom of the decanter through the annualar space around the upper-layer outlet tube. The overall level is maintained slightly above the level of the coolant outlet port. The height difference between the overall liquid level and the top of the upper layer outlet tube provides room for process control of the overall level. By controlling the outlet flowrate of the lower layer, the interface level can be maintained, whereas controlling the outlet flowrate of the upper layer maintains the overall liquid level.
Due to the two 60-mm Schott Duran flanges, the design of the decanter is very modular. If the diameter or height of the decanter needs to be changed, the decanter body can be replaced. The upper-layer outlet tube can be easily replaced with a different length. The feed tube length can be modified by a glass blower by removing the upper glass piece. Glass will accommodate acidic liquids and, as mentioned earlier, the guided-wave radar rod can be replaced with an exotic metal.
A consideration that affects how the decanter is connected to the column is that of which liquid phase is refluxed to the column. If both liquid phases are refluxed in equal proportions, it may be possible to use a liquid dividing head to split the condensate for reflux and send the distillate to a decanter, although this can be tricky if there is strong phase separation between the liquids. If only one liquid phase will be refluxed, then the condensate should be decanted first and the refluxed layer can then be sent to the reflux splitter. For the cases in which both phases need to be refluxed, but at different ratios, the condensate should first be decanted, then each phase should refluxed separately (separate reflux splitter or pumps).
Polymer formation
Polymer formation is a special problem for distillation columns. Some examples of chemicals that easily polymerize are acrylates, acrolein, acrylonitrile, acrylic acid and styrene. The difficulties posed by these compounds are associated with how easily they polymerize, the solubility of the polymer in the monomer, and how difficult it is to inhibit the polymerization.
Examples of polymerization inhibitors are phenothiazine, hydroxyquinone, quinone, and 4-hydroxytempo. Oxygen itself hinders polymerization, but it also activates some inhibitors. Metal surfaces, such as copper and Monel alloys, also hinder polymerization.
The inhibitors listed above have low vapor pressures and essentially do not exist in the vapor phase in a distillation column, so vapors condensing on uninhibited surfaces are prone to polymerization. The two main areas of a distillation column that require special attention, due to this process, are the condenser and the column.
Condenser. It is important that the cooling surface of the condenser is coated with inhibited liquid. Two of the ways to accomplish this are with the use of a Friedrich condenser for laboratory columns and a spray condenser for pilot columns.
Figure 11 shows an example of a typical Friedrich downdraft condenser. The cooling surface is a spriral inner tube with close tolerances between the spirals and the outer tube. Inhibitor solution introduced at the top tends to spiral down and coat this cooling surface. These condensers works well for small columns, but lack enough cooling surface and vapor-flow capacity for larger columns. The advantage is that this solution is simple.
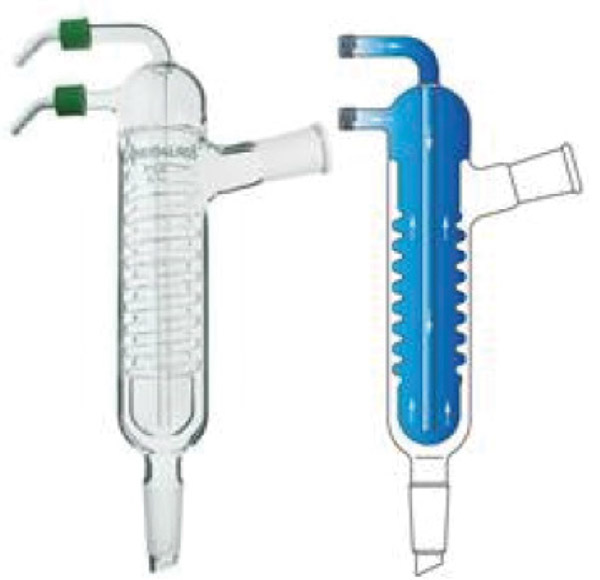
FIGURE 11. Friedrich condensers may be necessary to insulate the condenser to avoid condensation on the outer wall (photo and drawing used with permission from Chemglass Life Sciences)
An enhanced Friedrich-style downdraft condenser is shown in Figure 12. This condenser is constructed of three spiral Friedrich sections fused together for increased surface area. Inhibitor is fed to the top of the upper Friedrich section and the vapor inlet is halfway down this section. This allows the inhibitor to coat the surfaces above the vapor inlet to minimize the possibility of having uninhibited condensation surfaces, even if some of the vapor flows upward and condenses. In addition, the condenser has a jacket which can be evacuated to full vacuum or heated. This minimizes or eliminates any condensation on the outer process wall, in case this surface is not well-coated with inhibitor.

FIGURE 12. The condenser shown here has three Friedrich sections. In addition to increased surface area, it also has a jacket that can be evacuated for insulation and enhancements for inhibitor introduction. This jacket prevents condensation on the wall, which may not have an inhibitor (Photo used with permission from H.S. Martin, part of the AGI Group)
Figure 13 shows an example of a spray condenser. The condensate is pumped around in a loop, through a heat exchanger, for heat removal, and a spray nozzle located inside of a collection vessel. Inhibitor is introduced into the recirculation loop. The vapor from the column is condensed by the spray, which also ensures that all of the surfaces are coated with inhibitor.
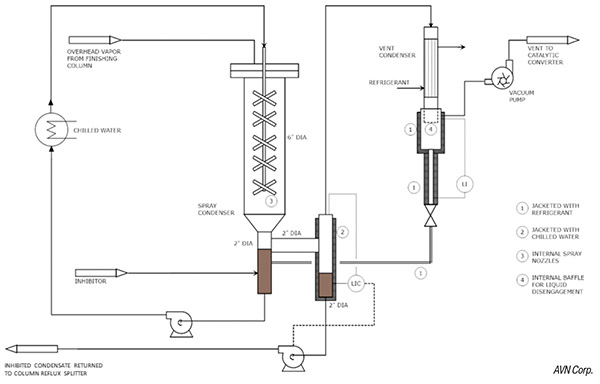
FIGURE 13. The diagram shows a spray condenser system
Column. Trays work well for polymerizable chemicals because the inhibited liquid spray tends to coat most of the column walls and other surfaces. Packing lacks this spray feature and has more of a tendency to have uninhibited surfaces on the column wall and within the packing. Packing can be used in certain cases — for example, if the polymer has at least some solubility in the monomer, but in general, has more tendency to foul due to polymer formation.
Figure 14 shows a custom Oldershaw column section designed specifically for polymerizable chemicals. It has Schott Duran O-ring joints that seal well and allow the column section to be removed with minimal vertical movement of the adjacent sections. Special heating mantles are wrapped around the joints to eliminate heat loss, condensation and potential surface polymerization.
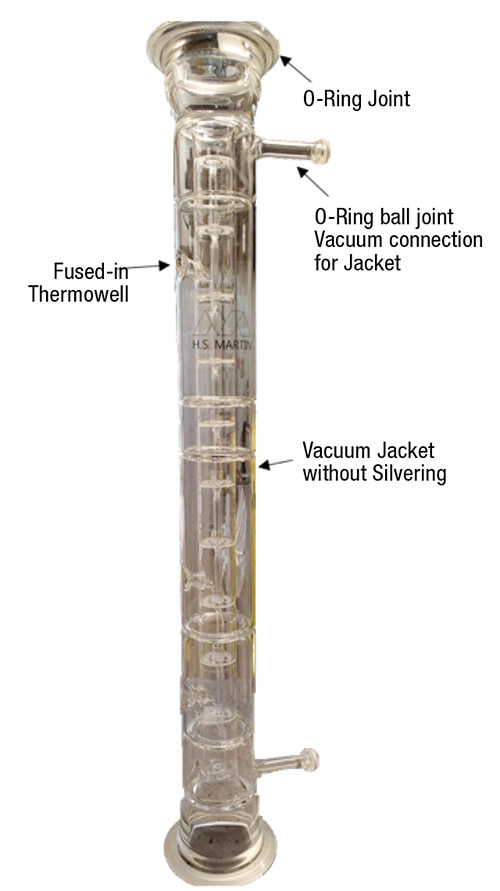
FIGURE 14. This Oldershaw column section is designed for polymerizable chemicals. It has an unsilvered jacket that can be evacuated with a vacuum pump, fused-in thermowells, and O-ring joints that have heated mantles to prevent heat loss (photo used with permission from H.S. Martin, part of the AGI Group)
Although most vacuum-jacketed glass Oldershaw column sections are silvered to prevent radiant heat loss, the column section show in Figure 14 was designed for temperatures that do not exceed 80˚C, at which radiant heat losses are minimal. The jacket can be connected to a vacuum pump to eliminate heat loss by convection. With this arrangement, if polymerization does occur on the trays, the column section can be easily removed and placed into an annealing oven to be baked out. This treatment on a traditional silvered column section would destroy the silvering and vacuum. In this case, the column would need to have the vacuum broken, baked out with silvering removed, re-silvered, and have vacuum resealing, which is significantly more time consuming and expensive.
Traditionally, tray temperature measurements involve either thermocouples inserted directly onto the tray or removable thermowells installed just above the tray. In either case, ball joint connections are usually used for the thermowells. However, this arrangement tends to have cold spots in which condensation can occur. If the chemicals are polymerizable, the condensation may occur on uninhibited surfaces, leading to polymerization, possibly gluing the thermowell or thermocouple to the column section permanently. The column section in Figure 14 has fused-in thermowells that eliminate this issue. Fused-in thermowells can also be used for high-vacuum service (<50 mm Hg), where we wish to minimize air leaks into the system. It is important when using fused-in thermowells that the thermowell is filled with oil or a thermal paste for good heat transfer.
Edited by Scott Jenkins
References
1. Oldershaw, C. F., Perforated Plate Columns for Analytical Batch Distillations. Industrial & Engineering Chemistry Analytical Edition, pp. 265–268, 1941.
2. Kister, H. Z., “Distillation Design,” McGraw-Hill, New York, N.Y. 1992.
3. Lewis, W. K., Rectification of Binary Mixtures. Ind. Eng. Chem., pp. 399–402, 1936.
4. Performance Distillation Solutions; spun off from Cannon Instrument Co. (n.d.). Pro-Pak Protruded Metal Distillation Packing Literature. State College, Pa.
5. Koch-Glitsch. (n.d.). Structured Packing. Retrieved from https://koch-glitsch.com/technical-documents/brochures/structured-packing-brochure.
6. Sulzer. (n.d.). Laboratory Packings. Retrieved from https://www.sulzer.com/en/shared/products/laboratory-packings
7. Krell, E., “Handbook of Laboratory Distillation,” Elsevier, Amsterdam, the Netherlands, 1982.
8. Frank, T. C., Liquid-Liquid Extraction and Other Liquid-Liquid Operations and Equipment. In D. W. Green (Ed.), “Perry’s Chemical Engineer’s Handbook,” 9th ed., pp. 15–87, McGraw-Hill Education, New York, N.Y., 2019.
Authors
 Glenn Graham is a corporate fellow at AVN Corp. (which recently acquired Matric; 1740 Union Carbide Drive, South Charleston, WV 25303; Email: glenn.graham@AVNcorp.com; Phone: 304-552-6554). Graham is responsible for providing technical guidance and support for laboratory- and pilot-scale research and development projects, including ASPEN computer simulations, the design and operation of laboratory- and pilot-scale equipment, and experimental data analyses. He specializes in distillation and related technologies. He joined AVN in 2013. Prior to joining AVN, Graham worked for Union Carbide and Dow Chemical in their R&D separations groups, also specializing in distillation and related technologies. Graham holds B.S.Ch.E and M.S.Ch.E degrees from Montana State University.
Glenn Graham is a corporate fellow at AVN Corp. (which recently acquired Matric; 1740 Union Carbide Drive, South Charleston, WV 25303; Email: glenn.graham@AVNcorp.com; Phone: 304-552-6554). Graham is responsible for providing technical guidance and support for laboratory- and pilot-scale research and development projects, including ASPEN computer simulations, the design and operation of laboratory- and pilot-scale equipment, and experimental data analyses. He specializes in distillation and related technologies. He joined AVN in 2013. Prior to joining AVN, Graham worked for Union Carbide and Dow Chemical in their R&D separations groups, also specializing in distillation and related technologies. Graham holds B.S.Ch.E and M.S.Ch.E degrees from Montana State University.
 Raymond Rooks is a principal engineer at AVN Corp. (formerly Matric; same address as above; Email: raymond.rooks@AVNcorp.com; Phone: 304-720-1037). Rooks has more than 20 years of experience in the chemical industry. He has held positions at Simulation Sciences, Union Carbide/Dow, and Praxair/Linde. He has broad experience in process development, including process modeling, simulation, experimental design, and experimental/laboratory simulation, especially in process separations. Rooks holds a Ph.D. in chemical engineering from the University of Massachusetts at Amherst and a B.S.Ch.E. from the University of Oklahoma.
Raymond Rooks is a principal engineer at AVN Corp. (formerly Matric; same address as above; Email: raymond.rooks@AVNcorp.com; Phone: 304-720-1037). Rooks has more than 20 years of experience in the chemical industry. He has held positions at Simulation Sciences, Union Carbide/Dow, and Praxair/Linde. He has broad experience in process development, including process modeling, simulation, experimental design, and experimental/laboratory simulation, especially in process separations. Rooks holds a Ph.D. in chemical engineering from the University of Massachusetts at Amherst and a B.S.Ch.E. from the University of Oklahoma.