
By focusing on ignition sources, such as static discharges, and housekeeping, facilities handling solids can minimize their risk for combustible dust explosions
Dust explosions in industrial facilities can cause injuries, destruction of property, and — as history will attest — unfortunately, fatalities. A U.S. Chemical Safety and Hazard Investigation Board (CSB; Washington, D.C.; www.csb.gov) report conducted in 2006 identified 281 combustible dust incidents between 1980 and 2005 that killed 119 workers with injuries sustained to 718 others, along with several damaged facilities. In the U.K., the government agency Health and Safety Executive (Merseyside, U.K.; www.hse.gov.uk) recorded 303 dust explosions over a nine-year period, and German records demonstrate 426 similar incidents over a 20-year period. Unfortunately, tragic incidents like these continue to cause significant business interruptions and loss of lives.
Given that such hazards exist within industry today, it is critical for plants to focus on areas where they significantly reduce the risk of dust explosions. These include close attention to good housekeeping practices, strict adherence to maintenance practices, and the identification and elimination of ignition risks for combustible dusts. This article provides information on the potential causes of dust explosions and on methods to minimize the risk of such incidents occurring.
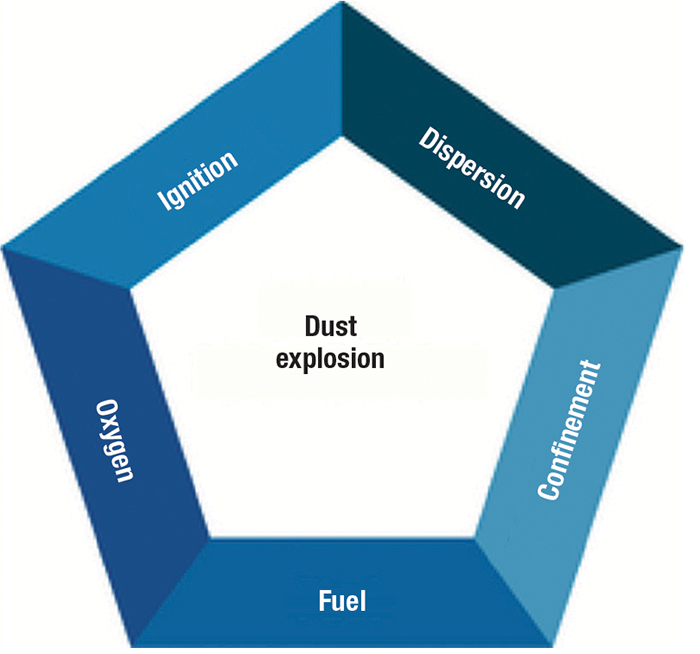
Dust ‘pentagon’
Dust explosions within hazardous-area processing environments are not a new danger, nor is static charge being a potential source of ignition. The earliest recorded dust explosion was at Giacomelli’s Bakery in Turin, Italy in 1785, where flour dust generated during normal operations came into contact with a mounted lamp. Flour can become combustible if it is too dry and builds up a static charge. The bakery owner was recorded as saying that the flour was the driest seen in the bakery that year. A dispersed cloud of flour originated when flour from the upper portion of the warehouse dropped to the confined warehouse below. The resulting explosion that followed injured both the worker shovelling flour into an open flame and a boy who fell from scaffolding, as a result of the flames blowing out windows onto the street. What this incident inadvertently demonstrated, apart from the violent and volatile nature of a dust explosion, was an archetypal insight into the “dust explosion pentagon.”
Five elements are required to intiate a dust explosion. Beyond the fire triangle (fuel, oxygen, heat source), a dust explosion requires two additional elements, in the form of dispersion of dust particles in the right concentration and the confinement of the dust cloud itself (Figure 1). Dispersed airborne dust burns more rapidly, and confinement allows for pressure build up. Having these two additional elements in place, along with the three elements of the original fire triangle, increases the likelihood of a dust explosion.
By definition, a combustible dust is any fine material that has the ability to catch fire and explode when dispersed in air. By their very nature, dust explosions emit an energetic force, creating powerful pressure waves that can cause significant harm to personnel and damage to facility structures and equipment. Dust explosions can also generate intense heat inside the dust cloud and can cause structures to fall, creating additional risks for plant personnel.
Areas of risk
Dust is a hazardous byproduct in many industries in which handling powders is involved. This includes companies engaged in a wide range of sophisticated manufacturing processes. Dust explosions can occur in any industry handling combustible dusts, including (but not limited to) the following:
- Coal
- Wood
- Waste recycling (paper)
- Agriculture
- Chemicals
- Metal processing
Over half of dust explosion incidents can be attributed to one of these four industry sectors: food products, wood products, chemicals and metals. The average dollar loss per explosion incident in a typical year is $3.4 million.
The National Fire Protection Association (NFPA; Quincy, Mass.; www.nfpa.org) Standard 654 (Standard for the Prevention of Fire and Dust Explosions from the Manufacturing, Processing, and Handling of Combustible Particulate Solids) defines dust as “A combustible particulate solid that presents a fire or deflagration hazard when suspended in air or some other oxidizing medium over a range of concentrations, regardless of particle size or shape.” Many organizations are uninformed and unaware that their processes can produce an atmosphere that can be explosive, and as a result, dust can be produced by a seemingly innocuous consequence of their usual manufacturing process.
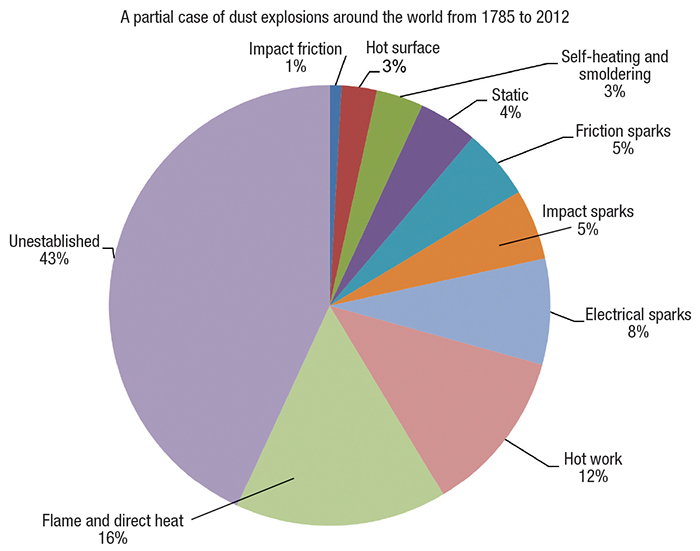
Figure 2. A wide range of ignition sources exist for dust explosions in process plants
There are a number of primary ignition sources that can trigger a dust explosion (Figure 2), including the following:
- Electrostatic ignition
- Friction
- Electrical arcing
- Hot surfaces
- Open flame
- Self-ignition
Particle size
Dust explosions occur when a dispersed combustible material is present in a sufficiently high concentration. Therefore, an explosion hazard exists whenever dusts are produced, stored or processed, and whenever these materials become airborne. When the product being processed is considered combustible and has an appreciable portion of fine material, the potential for having an explosion increases dramatically. Fine powders with low minimum ignition energies (MIE) will regularly reach the minimum explosive concentration (MEC) along conveying systems and may be at risk of combustion by several sources of ignition. One such ignition source is electrostatic discharge.
The MIE required to ignite any given powder depends on the fineness of particles, with the lowest MIE values tending to relate to very fine particles (Table 1). In situations where the MIE for a dust material is above 10 Joules, and no flammable gases or vapors are present, taking special measures to minimize static electricity is usually not necessary. However, precautions could still be necessary with conductive plant equipment capable of storing high levels of static charge, to minimize electric-shock risks for operators. In the chemical and pharmaceutical industries, MIE values required for ignition are often relatively low (especially in flammable dust-vapor hybrid atmospheres), while in the food industry, MIEs are usually somewhat higher.
The ease of ignition and severity of combustible dust explosions are typically influenced by particle size. Finer particles are more explosive than coarse particles because they have larger surface areas relative to their weight, allowing them to rapidly react with oxygen when dispersed in air and ignite (Table 2).
In NFPA Standard 654, dusts are defined as solid particles with particle diameters of 420 microns (µm) or smaller. For perspective, the particle size of table salt is around 100 microns.
Primary and secondary explosions
One of the main dangers of a dust explosion occurs when combustible dusts ignite and cause a chain reaction with other dusts. Dust explosions create their own self-sustaining domino effect, which continues as long as fuel exists. A secondary explosion occurs when dust accumulated on floors or other surfaces is lifted into the air and ignited by the primary explosion. The primary explosion liberates dust from surrounding surfaces (including beams and ledges), causing them to be suspended in air and dispersed. The secondary explosion propagates from the resulting dust cloud. Secondary explosions occurring when the structural integrity of the facility may already be compromised can result in catastrophic results.
Primary and secondary explosions often occur together, allowing for transitions from fire to multiple explosions and vice versa. In many cases, a primary explosion occurs inside a process vessel, such as a sieve, dryer, mixer, conveying system or silo (Figure 3). It is within these confined environments that the five elements of the dust explosion pentagon are met. The resulting impact of the first explosion usually ignites the second. Unburned fuel from the primary explosion is ejected by the blast outside the enclosure, causing dust to become airborne, where it is susceptible to ignition and is capable of triggering a secondary explosion.

Figure 3. Dust explosions often have primary explosions, which occur in confined process equipment, and secondary explosions, which ignite material dispersed by the initial explosion
A secondary explosion can be more destructive than the primary due to the increased concentration of dispersed combustible dust and stronger ignition source. The resulting shockwave of an initial explosion will damage and often rupture the self-contained vessel where the initial blast occurred, allowing the explosion to propagate through the plant.
Imperial Sugar explosion
The explosion at the Imperial Sugar refinery in Port Wentworth, Georgia in 2008 provides and illustrative example of a dust explosion and highlights the devastating effects that any dust explosion can have on people, businesses, communities and the economic climate (Figures 4 and 5). The incident claimed the lives of 14 workers and critically injured 36 others. A year before the blast, in 2007, the company had produced 1.3 million tons of sugar, making it one of the largest sugar refineries in the U.S. A series of violent sugar dust explosions obliterated the site, with 12% of the 160-acre facility completely destroyed. According to the subsequent CSB investigation, all 14 casualties from the blast were “most likely the result of the secondary explosions and fires.”

Prior to this destructive incident, the Port Wentworth facility had operated for more than 80 years without experiencing a devastating dust explosion. However, the plant had a history of long-standing problems of sugar dust and spilled sugar in the packing buildings and silo penthouse between 1970 and 2007. It is likely that, although conditions suggested that an incident should occur, the sugar dust never accumulated to levels above the MEC before the event. It seems to have been just fortune that dust from spilled sugar never reached levels where the concentration was high enough for an explosion to propagate through a plant until the incident. Internal correspondence documented the amount of spilled sugar as “knee deep” in some areas of the plant. Two months prior to the incident, an internal inspection showed that tons of sugar were accumulating on the floor of the facility. This provided much of the fuel for the secondary explosions.

Figure 5. The Imperial Sugar explosion resulted in 14 fatalities, most of which occurred during the secondary explosion and fire
At the Port Wentworth site, granulated sugar was stored in three 100-ft silos and then conveyed into packing buildings, where it was packaged for distribution. The primary dust explosion occurred inside the enclosed steel conveyor belt used to transport granulated sugar under silos 1 and 2. Airborne combustible sugar dust accumulated above the minimum explosible concentration inside the unit, and this triggered huge secondary explosions and fire throughout the packing buildings. In the aftermath of the incident, the CSB reported that for many years, granulated sugar on these conveyors was exposed to possible contamination from debris that could fall into the sugar. As a result, Imperial Sugar installed a stainless-steel frame to enclose each belt assembly to protect granulated sugar from debris and contamination. These panels were able to be removed for cleaning operations, but were not equipped with a dust-removal or explosion-venting system. The stainless-steel enclosures essentially confined the sugar such that sugar dust was able to accumulate to a potentially explosive concentration above the MEC within the conveyor. An overheated bearing was determined to be the likely ignition source, and the explosion was triggered in that way. In addition, multiple other potential dust-ignition sources were identified inside the enclosure.
Imperial Sugar CEO John Sheptor stated at the time that “accumulated sugar likely acted like gun powder,” and called combustible dust “a silent risk that needs to be addressed.” In the 1925 book “The Dust Hazard in Industry,” author William Gibbs concluded that “Sugar, dextrin, starch and cocoa are the most dangerous,” in terms of dust explosions in industrial facilities, with sugar being exceptionally so. “Sugar ignites when projected as a cloud against a surface heated to below red heat, and when ignition has taken place, the flame travels throughout the dust-cloud with great rapidity,” Gibbs wrote. In the wake of the Imperial Sugar explosion, The Occupational Safety and Health Administration (OSHA; Washington, D.C.; www.osha.gov) cited the company for 108 instances of “willful violations related to the combustible dust hazard, including the failure to clean up spilled dust and not using appropriate equipment or safeguards where combustible dust is present.” At the time, the proposed fine of $8.7 million for the sugar refinery explosion was the third largest in history for an industrial safety incident.

Preventing static discharge
In manufacturing and handling process industries involving flammable and combustible atmospheres, the threat of static electricity is ever present. There are certain types of dust-handling plant equipment in which static electricity is readily generated. These include mills, conveyor belts and pneumatic conveying systems. In potentially explosive atmospheres, the amount of energy contained in spark discharges from plant equipment, and even from people, may be sufficient to ignite many fine dusts produced during handling of loose solids, such as powder, granules, pellets and flakes. Electrostatic charging of isolated plant equipment or materials is likely when moving dusty materials in quantity. It is fundamentally critical to take necessary precautions to prevent discharges that are powerful to cause ignition of a dust cloud.
All potential sources of internal and external static discharges from process equipment situated in zoned and classified areas must be accounted for and managed in the appropriate way. If they are not sufficiently bonded and grounded, isolated components in conveying and dust collection systems are capable of holding large amounts of static electricity. Isolated components usually result from design oversight, or after maintenance teams reassemble fittings without re-establishing static bonding connections. Pipes, valves, blowers, hoppers and other components engaged in powder transfer processes can be isolated from each other due to the insulating properties of parts like rubber gaskets, or through general wear and tear. The most secure means of preventing charge build-up is to bond and ground components to a reliable verified earth.
Although the generation and accumulation of static electricity is invisible and discrete, it holds a very real possibility of a potential discharge and the risk of igniting combustible atmospheres. To prevent electrostatic discharges from igniting combustible dusts, companies should conduct risk assessments of their processes and equipment to ensure all potential sources of static ignition are identified and managed correctly.
Each explosion threat presents its own unique set of challenges. The variables involved, from the combustible material, ignition source, process vessel, operational procedures and environmental conditions, all impact the degree of risk and the severity of the consequences of an explosion. There are a number of practical solutions to consider in order to safeguard the plant, people and processes, such as grounding and bonding, explosion venting, suppression and isolation, among others. Static grounding and bonding systems are designed to eliminate static as an ignition source before combustible material is allowed to ignite. In safety terms relative to the dust fire pentagon, removing the ignition source eliminates one of the key elements required for an ignition.
Despite the potential for extensive loss of human life, damage to facilities and downtime in production, history has shown that necessary actions and preventive measures have not always been taken. Of course, it is not always possible to completely eliminate the risk of dust-related explosions, but the prevention is critical.
The nature of a powder processing operation means that the generation of static electricity is to be expected in all parts of the system because of the movement of the particles through equipment. Therefore, regular maintenance is required to stop material from clogging up the machinery. Regular disassembly for cleaning and maintenance can result in bonding connections being missed or not made correctly when the equipment is reassembled. Vibration and corrosion may also degrade assembly connections, so it is imperative to ensure that no parts in the assembly become isolated from a true earth ground. The most effective way of ensuring that the complex equipment used in powder-processing operations cannot accumulate static electricity is to provide a dedicated static grounding solution that is capable of monitoring the ground connection to components at risk of isolation (Figure 6). Such a solution is able to prevent the flow of product and alert personnel to a potential hazard should a component lose its connection to ground. This is especially important if the ground connection point to the equipment is not readily visible, or isn’t easily accessible (for example, grounding clips). Powder-processing equipment presents more of a challenge compared to standard applications, because there are metal parts that can make up larger assemblies that can be electrically isolated from each other. The risk of removable sections becoming isolated conductors will occur if each section does not have a sufficiently low path to ground to safely dissipate charge, and also if the correct reassembly of equipment after cleaning, between operations and regular examination of bonding straps between the metal pipework and duct sections by plant personnel is not routinely carried out.

Figure 6. A static-electricity grounding system can reduce the risk of dust explosions. In the conveying system shown above, the numbers represent parts of the system that could be at risk for isolation

Other prevention concepts
Additional areas of consideration to prevent dust cloud accumulation in solids-handling facilities include the following:
- Maintain a plant in a leak-tight condition (fix damaged seals, tighten loose bolts)
- Reduce labor involved in cleaning by designing plants within the minimum number of horizontal ledges where dust can settle
- Electrical plant equipment may be at risk of overheating if dust deposits settle
- Introduce technical measures to safeguard against an explosion
One of the most important risk-mitigation measures is maintaining a clean working environment. If dust deposits are allowed to accumulate, they can provide fuel for a secondary explosion.
To prevent uncontrolled static discharges posing a fire and explosion hazard in powder-processing operations, a thorough static audit conducted by qualified personnel should be carried out. For situations where potentially isolated components are identified, dedicated grounding equipment should be installed to monitor and control the release of static electricity, thereby removing a primary source of ignition in combustible dust atmospheres.
Given that such hazards exist within industry today, good housekeeping, strict maintenance practices, and ignition risk identification and elimination are paramount in preventing a dust explosion.
Edited by Scott Jenkins
Author
 James Grimshaw is the marketing manager of Newson Gale Ltd. (Omega House, Private Road 8, Colwick, Nottingham, NG4 2JX; Phone: +44 (0)115 940 7537; Email: james.grimshaw@hoerbiger.com; Website: www.newson-gale.co.uk) Newson Gale is part of the Hoerbiger group. Grimshaw’s background and experience within the business of safety stems from working in business-to-business environments with organizations that provide technologies used to protect assets and its people.
James Grimshaw is the marketing manager of Newson Gale Ltd. (Omega House, Private Road 8, Colwick, Nottingham, NG4 2JX; Phone: +44 (0)115 940 7537; Email: james.grimshaw@hoerbiger.com; Website: www.newson-gale.co.uk) Newson Gale is part of the Hoerbiger group. Grimshaw’s background and experience within the business of safety stems from working in business-to-business environments with organizations that provide technologies used to protect assets and its people.