Commonly used in the CPI, sulfuric acid requires many special precautions to ensure its safe handling and storage

Figure 1. Storage tanks in sulfuric acid service require many special precautions to ensure safe operations and prevent accidental spills or ignitions
Sulfuric acid, sometimes called the “king of all chemicals,” is widely used in the chemical process industries (CPI) for the manufacture of various fertilizers and other chemicals. Sulfuric acid (H2SO4) is typically stored and handled in steel storage tanks in tank farms (Figure 1) located at ports, sulfuric acid plants, fertilizer plants and so on. Numerous incidents involving explosions and spills in sulfuric acid storage tanks have been reported worldwide. All too often, a lack of understanding regarding the proper safety aspects required for handling and storing sulfuric acid leads to catastrophic accidents. These incidents can result in environmental pollution, as well as injuries and fatalities.
For example, in 2001 at a petroleum refinery in Delaware, a crew of contractors was repairing grating on a catwalk in a sulfuric acid storage-tank farm when a spark from their hot work ignited flammable vapors in one of the storage tanks. One of the acid storage tanks separated from its floor, instantaneously releasing its contents. Other tanks in the tank farm also released their contents. A fire burned for approximately 30 min, and sulfuric acid reached a nearby river, resulting in significant environmental damage. One worker was killed and eight were injured [ 1 ].
Such incidents draw attention to the vulnerability of sulfuric acid storage tanks to fires and explosions, in addition to accidental spillage. These occurrences can be avoided by understanding the various aspects of corrosion in storage tanks, inspection and venting requirements, spillage-containment systems, instrumentation, proper materials of construction, various governing codes and standards, guidelines for hot work and so on. This article describes various problems faced in sulfuric acid tanks and the efforts needed to mitigate them.
Corrosion control
Corrosion is one of the most prevalent issues in handling sulfuric acid. Two common corrosion mechanisms — hydrogen grooving and boundary-layer corrosion — are especially dangerous in sulfuric acid service because they lead to the formation of hydrogen gas (H2), which is highly flammable, colorless, odorless and readily ignitable. It forms an explosive mixture with air and oxygen. The lower and upper explosive limits (LEL and UEL) of H2 are 4% and 74.2%, respectively. This means that if the concentration of H2 is between 4% and 74.2%, and if the gas mixture is ignited, it will lead to an explosion, causing potentially fatal harm and damage to assets. Hydrogen grooving and boundary-layer corrosion are detailed in the following sections. Hydrogen grooving. Tanks for storing concentrated sulfuric acid typically are made of carbon steel. The acid reacts with iron (Fe) in the shell, forming a protective layer of ferrous sulfate (FeSO4), while simultaneously releasing H2, according to the following reaction:
Fe + H2SO4 → FeSO4 + H2
At the inlet line, as the acid is flowing into the tank, H2 bubbles float up and come into contact with the metal. The bubbles will scrape off the iron sulfate coating, exposing bare metal. This will form more iron sulfate and release more H2 bubbles. Eventually, the formation of H2 bubbles in the metal forms grooves or corroded portions and is called hydrogen grooving.
Boundary layer corrosion. In tank farms, a corrosive atmosphere also prevails, as sulfur dioxide vapors from the storage tanks can combine with moisture, forming sulfurous acid (H2SO3). This causes the tank roofs and other iron-containing materials in the vicinity to corrode.
In acid plants, the sump tanks of absorption towers contain acid analyzers that control the addition of dilute acid. If these analyzers malfunction, it can lead to an increase in the acid temperature. If high-temperature acid is transferred to carbon-steel storage tanks, corrosion will occur at a much faster rate.
The general rate of shell corrosion of storage tanks for concentrated sulfuric acid service is approximately 5 to 20 mils per year (0.005 to 0.020 in./yr) [ 1 ]. Anodic protection should be provided for the storage tanks to minimize the corrosion of the shell.
Inspection
Periodic thickness measurements of a tank’s shell and roof should be carried out to ascertain whether the tank is fit for service. According to the recommendations of the National Association of Corrosion Engineers’ (NACE; Houston; www.nace.org) Standard RP 0294-94, an internal inspection of sulfuric acid tanks is to occur every five years, and an external in-service inspection is to be carried out every two years [ 2 ]. Similarly, the American Petroleum Institute (API) Standard 653 requires the evaluation of flaws, deterioration or other conditions that might affect the performance of a tank and the determination of its suitability for the intended service [ 3 ].
The periodic inspections should be performed more frequently as tanks become older. Per NACE RP 0294-94, inspection frequencies may be decided on the basis of operating conditions, experience, inspection results, fitness-for-service evaluations and risk analysis [ 2 ]. Furthermore, risk-based inspection (RBI) is to be conducted for all tanks in addition to inspections at service intervals. Historic tank leakage and failure data are integral to RBI assessments.
Instrumentation
As previously stated, sulfuric acid spills can be extremely dangerous. Therefore, level measurement and control in storage tanks in sulfuric acid service is of the utmost importance. First and foremost, all tanks should be provided with a level indicator. Additionally, high- and low-level switches should be provided on tanks to contain the overspill control and to avoid vacuum conditions when pumps are transferring acid to consuming plants. All instruments are expected to be compliant with safety integrity level (SIL) 2. For more information on SIL classifications, see Tolerable Risk, Chem. Eng., Sept. 2007, pp. 69–74.
For acid storage tanks, the use of radar-type level-measurement devices is recommended, along with control-room alarms. However, many facilities have installed differential-pressure (DP) level-measurement devices without any reported issues. The wetted parts of radar level gages should be 316 stainless steel, Alloy 20 or Teflon-lined.
Some older tank farms use a type of tank level-measurement device called a “bubbler system,” which uses instrument air as its medium. A small flow of instrument air (forming bubbles) enters the tank near its floor through the level probe. The air introduces some turbulence into the acid near the wall of the tank, which increases the corrosion rate by disturbing the protective FeSO4 layer. Instrument air also contains moisture and oxygen, much of which would likely be absorbed soon after coming into contact with the acid solution [ 1 ]. Hence, level measurements based on bubbler systems are to be avoided in sulfuric acid storage tanks, especially in spent-acid service, as air can form an explosive mixture with the volatile hydrocarbons present in the acid.
Flow measurement is also an integral part of sulfuric acid handling. Magnetic flowmeters, Coriolis mass flowmeters, rotameters or ultrasonic flowmeters may be used to measure the flow of acid into and out of tanks. As with level instruments, all flow instruments are to be SIL 2 compliant.
Switching of tank service
Some tanks must handle various forms of sulfuric acid, and switching between these materials introduces increased risk. For instance, in some tank farms, tanks must be emptied to switch material-handling service between oleum (an intermediate in sulfuric acid production) and sulfuric acid or between concentrated sulfuric acid and dilute spent acid. These switchovers do not typically require cleaning or draining, simply the removal of as much material as possible via pump. Each switchover represents an opportunity to exacerbate corrosion and potentially release sulfur trioxide (SO3) vapors or volatile hydrocarbon vapors.
Operators must be attentive and cautious with tanks in oleum service. Oleum is sulfuric acid (100%) containing around 23% free SO3. In some sulfuric acid plants, oleum is also produced by absorbing SO3 in concentrated sulfuric acid, and it is common practice to locate the oleum tanks near sulfuric acid tanks. When oleum is introduced in a storage tank, SO3 fumes are emitted out of the vent, creating visible pollution and a corrosive atmosphere. To curb the SO3 fumes, small-diameter packed columns filled with random packings (Intallox Saddles are recommended) are fitted on top of storage tanks, as shown in Figure 2. A bleed of sulfuric acid (98%) is taken from the transfer-pump discharge and fed to the top of the packed column to absorb the rising SO3 fumes from the tank. After absorbing SO3 fumes, the concentrated sulfuric acid is self-drained into the tank.
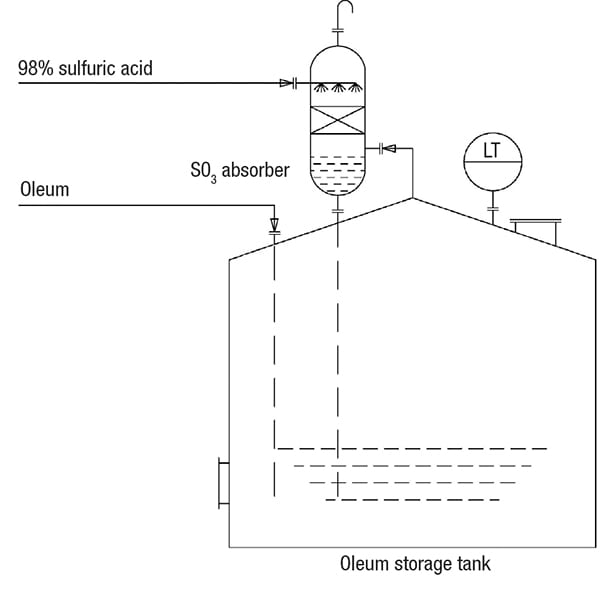
Figure 2. Oleum, a product of sulfuric acid plants, creates safety issues in storage tanks due to the presence of sulfur trioxide (SO3) fumes
Tanks in spent-acid service
Fresh sulfuric acid typically contains 98.4% acid and 1.6% water. Spent acid typically contains 88–95% acid and up to 5% water, with the balance consisting of hydrocarbons, including some light hydrocarbons that can vaporize. A spent-acid storage tank should be designed and operated as if it contained volatile hydrocarbons [ 4 ].
Special precautions are to be taken in storing spent sulfuric acid in storage tanks. The spent acid comes from various process industries as a byproduct and contains volatile hydrocarbons, which can be released during temperature variations in the atmosphere (thermal breathing). Therefore, blanketing of the storage tanks with an inert gas, such as carbon dioxide or nitrogen is advisable. Also, vent valves are to be provided with emergency tank venting and must be sized for the worst case, taking precautions for scenarios such as a failure of the inerting system (allowing excessive flow of inert gas to enter the tank) or an external fire that volatilizes hydrocarbon liquids in the tank. Additionally, flame arrestors should be placed in the vent line.
Tanks in spent-acid service should be provided with a frangible roof [ 1 ]. A frangible roof is a weak roof-to-shell attachment that preferentially fails over other welded joints when subject to overpressure. Failure of the roof-to-shell joint provides a means to relieve overpressure and to avoid catastrophic failure of the tank and loss of its contents.
Secondary containment systems
Special measures can be taken to ensure that no acid escapes to the environment in the form of secondary containment systems, including the installation of dikes. Dikes are to be built to contain 110% of the largest storage-tank capacity to contain the accidental overspill or catastrophic failure of the tanks. Dikes must also be designed to withstand not only the hydrostatic load of the liquid in the tank, but also for the dynamic tidal-wave effect of liquid-flow load in the case of tank failure. Acid-proof brick lining is to be laid inside the dike so that spilled acid does not permeate into groundwater. In addition to NACE RP 294-94 and API 653, API Standards 650 and 620 also provide helpful guidance in the construction of sulfuric acid storage tanks.
Although dikes can be an extremely helpful measure in protection against tank spills and failures, precaution must be taken when handling the rainwater that collects inside the dike area to prevent environmental contamination. Collected water from dikes in sulfuric acid service is acidic in nature because of the probable leaks of gases from vents inside the dike. This can create environmental issues if released to water bodies without correcting for pH. All rainwater that accumulates inside the dike area should be collected and tested for pH. If required, the pH should be adjusted to ensure it is within the acceptable range, typically 6.5 to 8.5.
Suitable valves must be placed at the dike wall so as to divert the clean rainwater to the dedicated rainwater drain and contaminated rainwater to an effluent treatment plant (ETP). Any accidentally spilled acid is to be collected in spill tanks and neutralized properly before disposal in the ETP.
PPE and HSE
Although sulfuric acid itself is not flammable, it should not be stored near organic materials, nitrates, carbides, chlorates or metal powders. Contact between high concentrations of sulfuric acid and these materials may cause ignition. Proper personal protective equipment (PPE) and health, safety and environmental (HSE) precautions are critical in any facility that handles sulfuric acid. Some best practices for PPE and HSE in sulfuric acid handling are as follows:
- Suitable eye-wash stations should be located near the tank farm for the safety of personnel. The eye washers are to be tested for their functionality every shift.
- All of the flanges in the lines are to be covered with lead cladding to protect the operating personnel from accidental acid leaks.
- Metal catwalks should be provided for working on top of tanks. Operating personnel should always use the catwalk and never walk directly on the tank shell (Figure 3).
- All lines should be sloped toward the storage tank, or toward the point of consumption. This will prevent the accumulation of acid in low points in the line, thereby eliminating possible safety hazards.
- Operating personnel should wear gloves that are loose enough to be easily removed in case acid enters them.
- Helmets should be worn at all times. To ensure eye and face protection, a full face shield and safety goggles are also necessary. Goggles and a face shield are especially crucial when working in a place where splashes can occur unexpectedly. It is essential that eye protection is provided from all angles.
- In the event of a small spill, one should contain and neutralize the acid with lime.
- Whenever a modification is carried out in the storage tank area, a hazard and operability (HAZOP) study should be conducted before proceeding to implementation.

Figure 3. A metal catwalk on the roof of a storage tank provides some protection for workers, as it is unsafe for personnel to walk directly on a tank’s shell
Venting requirements
Another crucial aspect to ensuring storage-tank safety is adequate venting. Some best practices associated with tank venting are as follows:
- Each sulfuric acid storage tank should be provided with an individual breather valve. The vent diameter is to be calculated based on in-breathing, out-breathing and thermal-breathing requirements according to API Standard 2000 [5].
- The vent area should be greater than the sum of the inlet-, outlet- and drain-line areas.
- The vent (breather) valve should be kept at the top of the tank. At no instance should it be located at ground level, as it has the potential for inundation in the case of tank rupture or the dike filling with acid or stormwater.
- If acid switchover (between either oleum and sulfuric acid or concentrated sulfuric acid and dilute spent acid) is routine practice in the tank farm, the vent valves must be sized for the worst-case scenario of vapor release.
- After decommissioning, the tank should be re-commissioned by introducing acid very slowly, so as to avoid static electricity.
- The structural integrity of storage-tank roofs is to be inspected in detail.
- An explosion-proof hatch cover (minimum of 600 mm in size) should be provided on the tank roof.
Transfer pumps
Transfer pumps are used to transfer sulfuric acid from the tank farm to the end-product processing plant or consuming plants (for example, a fertilizer plant), where acid is fed into chemical reactors. Careful attention must be paid to these pumps. Some recommendations for sound transfer-pump operations are as follows:
- The transfer pumps to the consuming plants shall have a low-level tank trip.
- The discharge line should be provided with a pressure gage and non- return valve.
- For the transfer of sulfuric acid from ships at a higher rate, the discharge line should have an excess-flow check valve to prevent accidental spillage of acid to the surrounding environment or to bodies of water.
Hot works in tank farms
Hot works — those tasks that require the use of flames or very high temperatures, such as welding and steel-cutting — must be completed with safety in mind. This is especially crucial in tank farms. The following are some guidelines for executing hot-works tasks in facilities where sulfuric acid is being stored:
- Hot work is to be permitted only after measuring for flammable gases in the storage tank area. The measurement of flammable gases in the storage tank area is to be done continually during the hot work period.
- According to API RP 2009, in situations where the work is delayed or suspended in an area that has previously been pronounced gas-free, the permit system should specify the length of time beyond which oxygen and flammability detector tests must be repeated or the permit reissued [6].
- Periodic combustible-gas and oxygen retests (or continuous monitoring) may be required while hot work is proceeding. The permit should specify the monitoring frequency.
- If the hot work is to be performed on the shell or roof of the storage tank, and if there are holes in the roof or shell, the tank contents must be emptied and purged with inert gas prior to the hot work.

FIGURE 4. Proper care must be taken when executing hot works in the presence of sulfuric acid storage tanks
In many instances, non-compliance with the above measures is a main cause for explosions in sulfuric acid tanks. Figures 4 and 5 illustrate the devastation caused by not taking the proper precautions with hot-works tasks. Figure 4 shows a storage tank in sulfuric acid service. Figure 5 shows the same tank after a catastrophic explosion due to the formation of H2 gas during hot work.
Pipelines, hoses and valves
Many special design and operating considerations should be taken for piping, hoses and valves that are to handle sulfuric acid. Table 1 provides some best practices for valves in sulfuric acid service. For acid-transfer lines, the pipeline should be of one piping class higher than required to mitigate the risk of accidental rupture or overpressurization.
The materials of construction for piping are dependent on fluid-flow velocities and quality concerns. If iron contamination is a concern for the process, as in the manufacture of caprolactam, carbon steel is to be avoided. Polyvinyl chloride (PVC) and chlorinated PVC are only to be used for vent lines and overflow lines. These pipes should not be used for liquid service. Table 2 provides some criteria for piping materials of construction based on velocity and temperature requirements.

FIGURE 5. The same tank from Figure 4 was badly damaged after its roof exploded due to hot work on the roof and the formation of hydrogen gas inside the tank while the tank was in service
In general, piping should be inspected visually every year. Ultrasonic thickness tests should occur biennially. Depending on the actual plant experience, an increase or decrease in the schedule of inspection may be required. Extra attention should be paid to elbows, tees, valves and any other areas in the piping where flow disturbances (and erosion or corrosion) could occur. Piping inspection criteria from API 570 may be used for guidance [ 7 ].
A hose lined with polytetrafluoroethylene (PTFE) is acceptable for 93–98% sulfuric acid service. The hose should be designed with a minimum working pressure of 14 kg/cm2 and be full-vacuum rated. The end fittings must be crimped or swaged. Banding is not recommended. The hose-end fittings should be 316 stainless steel with flanges or quick-connect fittings. The gaskets should be constructed of virgin Viton B materials. The user should have a hose-management program in place to ensure the integrity of hoses. All hoses must be dedicated to sulfuric acid service. [ 9 ]
Following the guidelines presented in this article will help enable engineers to safely approach the storage and handling of sulfuric acid. ■
Edited by Mary Page Bailey
References
1. U.S. Chemical Safety and Hazard Investigation Board (CSB), Investigation Report — Refinery Incident, Motiva Enterprises LLC, Delaware City Refinery, July 17, 2001.
2. National Association of Corrosion Engineers, NACE Standard RP 0294-94, Design, Fabrication and Inspection of Tanks for the Storage of Concentrated Sulfuric Acid and Oleum at Ambient Temperatures, 2006.
3. American Petroleum Institute, API 653 — Tank Inspection, Repair, Alteration and Reconstruction, November 2014.
4. McKetta, J.J., “Encyclopedia of Chemical Processing and Design,” CRC Press, June 1995.
5. American Petroleum Institute, API 2000 — Venting Atmospheric and Low-Pressure Storage Tanks, 7th Ed., March 2014.
6. American Petroleum Institute, API RP 2009 — Safe Welding, Cutting and Hot Work Practices in the Petroleum and Petrochemical Industries, February 2002.
7. American Petroleum Institute, API 570 — Piping Inspection Code, November 2009.
8.NorFalco LLC, Sulfuric Acid Handbook, www.myh2so4.norfalco.com/documents/NorFalco_ H2SO4TechBrochure.pdf, 2007.
9. DuPont Clean Technologies, Equipment Related Frequently Asked Questions, www2.dupont.com/Clean_Technologies/es_MX/assets/downloads/Equipment_FAQs.pdf.
Author
 Koya Venkata Reddy is senior manager, process engineering at FACT Engineering & Design Organization (FEDO), a division of Fertilizers And Chemicals Travancore Ltd. (FACT; Udyogamandal 683501, Kochi, Kerala, India; Phone: 91-484-2568763 Email: koyareddy@yahoo.com). He has 24 years of experience in chemical plant operations, including expertise in the fields of process control, process design, process risk analysis, HAZOP, process simulations, environmental management and plant revamps. He is a recipient of FACT’s Merit Award. Reddy holds a Bachelor of Technology degree from Andhra University, Visakhapatnam and a Master of Technology degree in project management from Cochin University of Science and Technology. He also received an M.B.A. in finance from Indira Gandhi National Open University (IGNOU), Delhi. He is a lifetime member of the Indian Institute of Chemical Engineers (IIChE) and a member of the Institution of Engineers (India).
Koya Venkata Reddy is senior manager, process engineering at FACT Engineering & Design Organization (FEDO), a division of Fertilizers And Chemicals Travancore Ltd. (FACT; Udyogamandal 683501, Kochi, Kerala, India; Phone: 91-484-2568763 Email: koyareddy@yahoo.com). He has 24 years of experience in chemical plant operations, including expertise in the fields of process control, process design, process risk analysis, HAZOP, process simulations, environmental management and plant revamps. He is a recipient of FACT’s Merit Award. Reddy holds a Bachelor of Technology degree from Andhra University, Visakhapatnam and a Master of Technology degree in project management from Cochin University of Science and Technology. He also received an M.B.A. in finance from Indira Gandhi National Open University (IGNOU), Delhi. He is a lifetime member of the Indian Institute of Chemical Engineers (IIChE) and a member of the Institution of Engineers (India).
