This column is based on “Acrylonitrile Butadiene Latex Production – Cost Analysis,” a report published by Intratec. It can be found at: www.intratec.us/analysis/nitrile-rubber-production-cost.
Nitrile rubber (also known as NBR, acrylonitrile butadiene rubber, acrylonitrile butadiene latex, Buna-N) is a synthetic polymer made of monomers 1,3-butadiene and acrylonitrile. This rubber is known for its resistance to chemicals, oils and fuels. NBR is produced with a wide range of molecular weights and monomer ratios, so basic polymer properties can vary greatly.
NBR is primarily used in applications where good oil resistance is required, such as rubber seals and O-rings, hose and belting stock and blowout preventers or packers (in oil drilling). NBR is also used in molding miscella-neous parts, rubber latex products, sponge applications, gaskets, pump stators, safety gloves and footwear. Additionally, NBR is used in modifying plastics, adhesives, cements, sealants, coatings and friction materials.
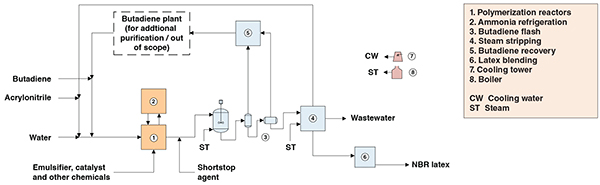
Figure 1. The diagram shows a process for the production of acrylonitrile butadiene latex
The process
The present analysis discusses an industrial process for acrylonitrile butadiene latex production. The process comprises two major sections: (1) polymerization; and (2) monomers recovery (Figure 1).
Polymerization. Polymerization occurs in continuously stirred, jacketed tank reactors connected in series. To maintain a low reaction temperature, all reactors are cooled by means of ammonia vaporization. Acrylonitrile and butadiene are mixed with an emulsifier, demineralized water and other chemicals to form the emulsion that will be fed to the polymerization reactors. Downstream, a “shortstop” agent is mixed with the emulsion in order to stop reaction at the desired conversion. This avoids gel formation.
Monomers recovery. The latex generated in polymerization reactors is flashed at atmospheric pressure and then under vacuum. Residual butadiene vapors are compressed and then cooled for condensation. The con-densed stream is directed to a decanter for water removal. Recovered butadiene is recycled to a butadiene plant, where butenes are separated, so the recovered butadiene can be reused in the NBR latex plant. The degassed latex is pumped and fed to a vacuum plate column, where residual acrylonitrile monomer is stripped by contacting the latex with steam entering the column bottom. Stripped acrylonitrile is recycled to the polymerization stage. The latex is mixed with an antioxidant agent and finally blended into a homogeneous emulsion before being sent to storage facilities as the finished latex product.
Production pathways
Nitrile rubber latex production basically consists of the emulsion polymerization of 1,3-butadiene and acrylonitrile, so different NBR manufacturing routes are related to different sources of the monomers used. The most typical NBR latex production routes are based on acrylonitrile produced via ammoxidation of propylene and butadiene produced via isolation from C4 steam-cracker fractions (Figure 2).
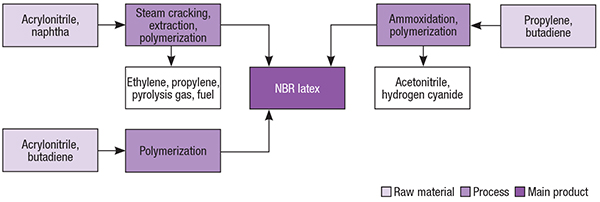
Figure 2. The most common NBR latex production pathways are shown here
Economic performance
The total operating cost (raw materials, utilities, fixed costs and depreciation costs) estimated to produce NBR latex was about $2,000 per ton of NBR latex in the first quarter of 2017. The analysis was based on a plant constructed in the U.S. with capacity to produce 35,000 metric tons per year of acrylonitrile butadiene latex.
Edited by Scott Jenkins
Editor’s note: The content for this column is supplied by Intratec Solutions LLC (Houston; www.intratec.us) and edited by Chemical Engineering. The analyses and models presented are prepared on the basis of publicly available and non-confidential information. The content represents the opinions of Intratec only. More information about the methodology for preparing analysis can be found, along with terms of use, at www.intratec.us/che.