Articles about thermal fluid systems often start with a variation of the statement that “thermal fluid systems typically require little ongoing maintenance for the first few years of operation” and then go on to extol the various advantages of indirect thermal-fluid process heating over competitive heating methods, such as direct heat, steam and so on. The corollary to that statement, however, is that by the time there is a problem, the operating personnel that were trained on the system have moved on, been excessed or promoted. As a result, when things do go wrong, the guessing begins. And, unless there is an obvious cause like a geyser from the expansion-tank vent or a pump that sounds like it’s moving ball bearings, someone will likely blame the thermal fluid for the problem.
There are several problems that seem to occur with some frequency. This article reviews a number of real examples and describes how the symptoms can be misinterpreted. The suspected fluid properties and the testing procedures necessary to determine which of the fluid properties (if any) is responsible for the problem are examined (Figure 1). Finally, recommended corrective actions are proposed.
Decrease in production rates
 |
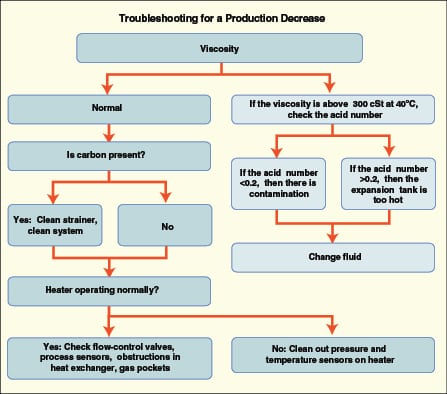
The following three examples explore production-rate problems, with the analyses and findings that resulted. A simple flowchart that can help in this type of analysis is given in Figure 2.
Example 1: A large facility experienced reduced productivity from its thermal fluid system that was operating at 450°F. The heater outlet temperature was increased to maintain throughput on several pieces of equipment, but the process was still losing ground. (Note: this is an excellent reason to log the heater outlet temperature so that you know when changes started, should you have a problem.) Previous test results had indicated the presence of carbon sludge in the fluid, so plans were made to activate a sidestream filter to remove the carbon. Before the plan was implemented, however, a sample of thermal fluid was taken that immediately identified the problem.
The fluid property that has the greatest effect on heat transfer rates is viscosity. Because the fluid heat-transfer coefficient (which controls the rate of heat transfer between the heat exchange surface and the fluid) is only one element of the overall heat-transfer coefficient, changes in the viscosity at elevated temperature (350°F or more) have to be significant (on the order of 200%) to cause a noticeable change in system performance. In this case, the problem was obvious and required no testing — a sample that was extracted at operating temperature went almost solid when it cooled. This put the fluid well above the 200% threshold.
Example 2: A chemical plant requested a sample kit to test its thermal fluid because one of its vacuum reactors was taking too long to heat. Even though the fluid had been in the system for many years and had recently been tested, it was assumed that the fluid must have gone “bad”. This situation is probably one of the more common scenarios for heat transfer systems. The problem comes to light when someone realizes that the heater temperature has to be increased to keep production on schedule. In this case, the evidence against the fluid was further strengthened by the relatively “normal” heater pressure and temperature readings. This prompted the request for the sample kit and a quotation for a complete fluid changeout. While the latter course of action is appealing to the fluid supplier, it was unlikely to solve the problem since the problem wasn’t the fluid.
The overriding evidence in this specific situation was that there had been no maintenance required on the system since the fluid had been tested. Fluid had not been removed or added (which eliminates contamination as a suspect) nor had any of the operating conditions changed. It turned out that there was a leak in the vacuum system, undetected by a faulty sensor, which resulted in an increased heat load required for distilling the product.
Example 3: A poultry processor was experiencing reduced throughput in a continuous convection oven. The heater and pumps were checked for problems, and all temperature and pressure sensors were replaced. Someone suggested cleaning the heat-transfer fluid system. Since the fluid had been in service for a number of years, it was assumed to have degraded and formed blockages in the coils because the temperature drop across the heat exchanger was much lower than when the unit was new. The fact that the fluid had been tested routinely and found to be in good condition was totally ignored in the evaluation. Management personnel wanted to clean the system and then change the heat transfer fluid. A lube-oil additive-type cleaner was added to the system with the expectation that the problem would be solved. When there was no progress, a thermal-fluid sample kit was requested along with a request to estimate the cost of replacement fluid. Once again the viscosity of the sample was found to be well within the normal range. The plant manager was very disappointed with the results showing that the fluid was not the problem, because he had to send the maintenance staff back in to keep looking for the real culprit. Eventually it was discovered that an air damper inside the oven had a broken weld that allowed it to flip up into the air stream, effectively blocking the coils. Throughput was reduced because insufficient heat was getting to that section of the oven.
Pressure fluctuations
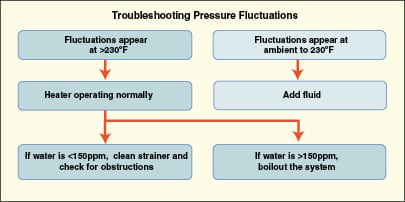
In one chemical plant, personnel noticed that the discharge pressure of the main circulating pump began to fluctuate as the fluid temperature approached 350°F after an extended shutdown. Thermal fluid was added to the system through the expansion tank, which made the situation worse for a period of time. Since the system had been kept under a nitrogen blanket during the downtime, water absorption through the expansion-tank vent was ruled out. Convinced that the fluid had degraded during the shutdown, personnel made plans to take another outage and replace the fluid. To pacify management, a fluid sample was taken and tested. The test results indicated high water levels (greater than 150 ppm, versus the normal level of less than 50 ppm).
Pump-discharge pressure fluctuations in a closed-loop heat-transfer system are always the result of entrained gas. Aeration of the fluid is often blamed for such fluctuations, particularly if fluid is added through the expansion tank. However, entrained air doesn’t abruptly become gaseous, but instead it causes problems from the start. While it is true that overheating a fluid can produce more volatile molecules that will theoretically vaporize, in practice the relatively low liquid-to-vapor expansion rate (which is about 20) pretty much rules this out as the source of gas.
The real culprit is most often water, which has an expansion rate of 1,000. Until water is either drained from the system or flashed off through the vent, it remains in the bottom of the thermal buffer tank or the expansion tank. In fact, tanks have been known to rust through at the bottom because water has been in the same place for many years. When the heat-transfer fluid flows out of the tank as the system cools, the water is carried into the system piping, and then is dispersed into the circulating fluid when the pump starts. As the system temperature reaches about 220°F, the water droplets become steam bubbles and the pressure fluctuations begin. What causes confusion is that the pressure problems don’t appear at the expected 212°F. Depending on the system pressure and design as well as the amount of water present, symptoms may not begin until the heater temperature reaches 280–300°F. If the pump is operating at a slightly negative suction head, even lower water concentrations can result in pressure fluctuations.
Figure 3 shows a simple chart that can be used to help troubleshoot pressure fluctuation problems.
Pump seals
A hot-roll calendering operation was experiencing repeated rotating joint-seal failures. The seal faces were being scored severely enough from the inside out to create fluid leakage. Two of the oldest seals were experiencing the greatest number of failures. While the fluid tests showed no significant change in the fluid condition, there was visible residue of a previous brand of insoluble fluid. The most compelling evidence of what the problem might be was that particles were collecting on the sidestream filter elements. To prevent further problems from what was suspected to be carbon (from degraded heat-transfer fluid), the user began to evaluate a system flush and fluid change.
The majority of carbon particles produced by fluid degradation are the result of fluid oxidation (as determined in fluid analysis by the total acid number). These acids are formed when hot fluid is exposed to air in the expansion tank. They are thermally unstable (compared to the fluid itself) and thereby degrade into carbon at relatively low temperatures (375 to 400°F) once the concentration has reached an acid number of 0.3 or so. If the expansion tank continues to run hot, the acid number will either stabilize or continue to increase. If the cause of the oxidation has been corrected, the acid number will decrease as the acids are consumed.
The carbon that is formed is similar to “soot” in appearance and will remain suspended in the fluid, which causes the fluid to appear to be black. The particles will drop out of suspension in stagnant fluid and form sediment (sludge). However, individually these particles are extremely fine (<0.5 micron) and as such are incapable of damaging rotating seals because they pass between the rotating faces. However, they will clump on 25-micron filter elements, which can be misleading during troubleshooting. In this case, the solution required analysis of the filter. The particles were analyzed and found to contain over 90% iron. This information was transmitted to the user, who then shelved the plan to flush the system and replace the fluid. Instead, he concentrated on improving his filtration system to eliminate the metallic particles.
Erratic production
A food processor began experiencing sporadic production problems with a multiple-user heat-transfer system that was used to heat tanks. Once again, the pump pressures and temperatures were all within the expected ranges. Because the fluid had been in service for a number of years, the likely solution was deemed to be fluid replacement. The shutdown was planned and quotes were obtained.
After the costs of the fluid and lost production were totaled, cooler heads prevailed, and it was decreed that the fluid should be tested by the current fluid suppliers to be sure it really did need to be replaced. Although the fluid had not been tested for a number of years and actually was a blend of several fluids, the supplier was able to determine that the fluid was in acceptable condition. Now that the “easy solution” was not applicable, the real investigation started. Particularly confusing, but overlooked when the fluid was the prime suspect, was the fact that the most significant decline in production occurred when there was the least demand on the heater. Fluid velocity has even more effect on heat transfer performance than viscosity, so whenever there is a drop in heat transfer, it’s time to look at the flowrate.
Liquid-phase heaters require continuous flow to prevent fluid degradation. Hence these systems need some way to bypass the heat users when heat is not required. There are two ways to accomplish this: 1) A backpressure control valve that maintains flow when the two-way control valves are closed; and 2) One or more three-way control valves (depending on the number of users) with a manual pressure-equalization valve on the bypass port.
Theoretically three-way valves are superior to a backpressure valve arrangement because they provide a constant flow through the heater — a concept that is favored by the purists — if the balancing is done rigorously. This exact balance is difficult to maintain over time due to changes in equipment and the ever-present potential for third-shift adjustments. In this particular case, it was discovered that the bypass valves on the least-used leg of the system had been fully opened so that when that system was not operating, a substantial amount of fluid was bypassing. When the unit was operating, the bypass volume was reduced, which in turn increased the pressure and thus flow to the other units bringing production rates back up. Instead of attempting to balance all of the bypass valves (which would have required the installation of multiple pressure gages) the solution was to install a backpressure valve between the feed and return header and then close all of the bypass valves, effectively turning them into two-way valves. While this control scheme did allow the heater flow to vary, it made the system much easier to control since each user was independent of the others.
Edited by Dorothy Lozowski
Author

Jim Oetinger is the director of technology at Paratherm Corp. (4 Portland Rd., West Conshohocken, PA 19428; Phone: 800-222-3611, Fax: 610-941-9191; Website: www.paratherm.com). He has over 30 years experience in the chemical and plastics industries. He has been involved with a wide range of products and processes including pigments, refrigerants, consumer plastic recycling, polymer compounding, process instrumentation and spray dried polymers. In addition, Oetinger has over 20 years experience in sales, marketing, and technical support of thermal fluids. He has authored articles on thermal fluid and system troubleshooting for this and other publications. A member of the Delaware Valley Chapter of the AIChE, he holds a B.S.Ch.E. from Clarkson University and a Masters of Management degree from Northwestern University. Oetinger and his family reside in a suburb of Philadelphia, Pa.