Several methods and calculations are available for determining particle size and particle-size distribution. This article provides and overview on how to use them
The behavior of particulate material is greatly dependent on its geometric properties, including particle size, shape and particle-size distribution (PSD). In industrial processes, these parameters affect phenomena such as sedimentation, flow through porous media, fluidization, gas-solid separation by cyclone, size enlargement (agglomeration) and powder mixing.
Similarly, particle size affects the end-use properties of many particulate products, such as the reactivity of catalysts, bioactivity and dissolution of drugs, setting time of cement, hiding power of cement, flowability of powder, and packing density of advanced materials. Particle properties also play a central role in the handling of solids-containing fluids.
Unfortunately, particle size characterization is among the most neglected areas in the field of chemical engineering, despite its importance in the vast majority of unit operations involving particulate matter and bulk solids handling. Since particle size measurement is routinely employed in many industries, appropriate techniques for particle measurement, and data presentation and interpretation, are of utmost importance to understand solids behavior, handling and final product qualities.
The measurement and selection of appropriate mean (average) particle size is a difficult task because of inherent particle characteristics. The difficulty is further compounded by the availability of a wide range of measurement techniques, which report different types of mean particle size and particle size distributions. The main objectives of this article are to address the following issues of particle characterization:
Describing single-particle size
Representing the size of a particle by its diameter is common, because the use of a single linear dimension to represent particle size is desirable for simplicity. Spherical particles can be conveniently defined by a single dimension (diameter). However, major issues arise for irregularly shaped particles, which is the case for most real-world applications.
There are several measures available to represent particle size, categorized into three areas (Figure 1, Table 1 and Ref. 1), as shown here:
(1) Equivalent spherical diameter
(2) Equivalent circle diameter
(3) Statistical diameters
Equivalent-spherical-diameter methods determine diameters by measuring a size-dependent property of the particle and relating it to a single linear dimension [2]. The equivalent sphere diameter takes advantage of the ideal shape of a sphere represented by the single dimension. The equivalent spherical diameter is the diameter of a sphere that shows the same controlling characteristics as the particle under investigation. The controlling characteristics could be volume, surface area, surface-area-to-volume ratio, settling velocity or other characteristics, as mentioned in Table 1, and Equation (1), which uses volume as the controlling characteristic. The equivalent volume sphere diameter (dv) equals the diameter of a sphere with the same volume as the particle (VP).
VP = π/6 d3v (1)
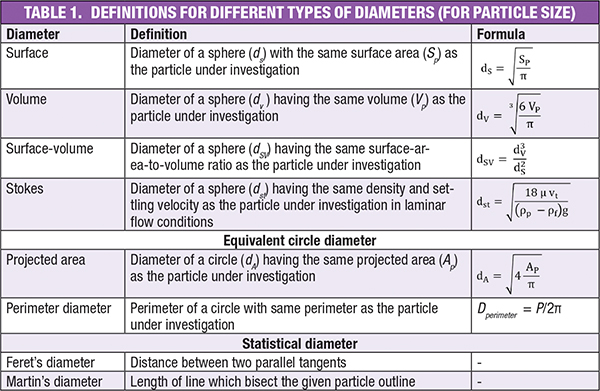
Hence, volume-equivalent sphere diameter is given by Equation (2).
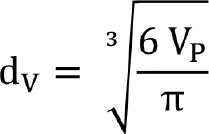 (2)
(2)
Several commonly used equivalent-sphere diameters are shown in Table 1 and Figure 1. More definitions can be found from Allen [ 2]. Microscopy is widely used to measure the particle size based on length.
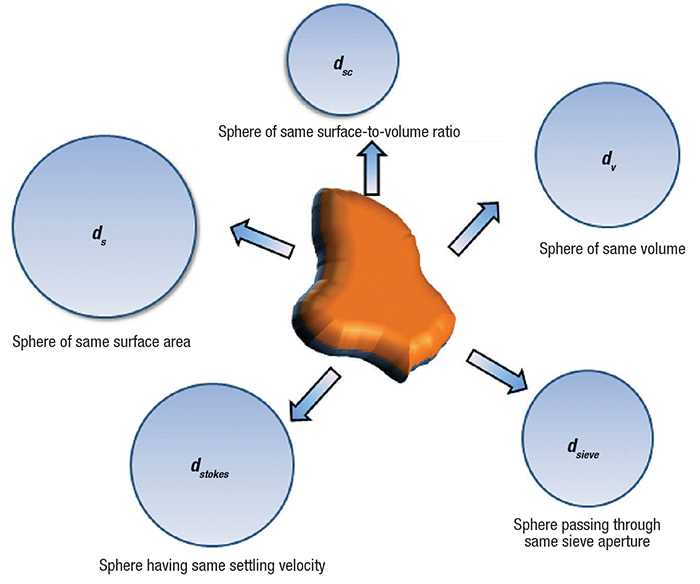
Figure 1. Equivalent spherical diameters based on different controlling characteristics can result in different particle size values for the same particle
The commonly used statistical diameters are: Feret’s diameter and Martin’s diameter. Feret’s diameter is defined as the distance between two parallel tangents, while Martin’s diameter is defined as a length of the chord that bisects the particle outline (Figure 2).
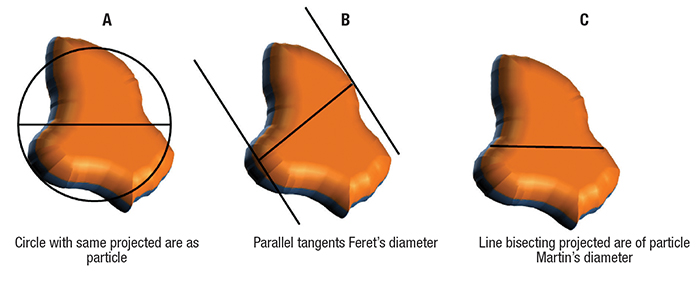
Figure 2. Martin’s diameter and Feret’s diameter are two types of statistical diameter from microscopy
Equivalent circle diameters, such as the projected area diameter (area of circle with the same area as the projected area of the particle under investigation), can also be used as shown in Figure 2A. These measures are outdated due to their statistical nature and poor reproducibility because there are many possibilities to estimate distance between tangents and bisector.
Size of an assembly of particles
Description of the mean (average) size of a population of particles is very challenging. It is advantageous to know the PSD of the solid products to establish the relationship between particle size and product performance. However, a single value for the mean particle size is often required for quality control and product comparison purposes. Hence, mean particle size is normally calculated from the given PSD. The average particle size represents the central tendency of PSD. The common measures of particle size for a given PSD are mean, mode and median (Figure 3). Mean size can be defined in different ways, including arithmetic mean, quadratic mean and Sauter mean. The mode (xmode) is the most commonly occurring size within the particle population and represents the peak of given PSD. The median size (x50%) represents particle size at 50% cumulative frequency.
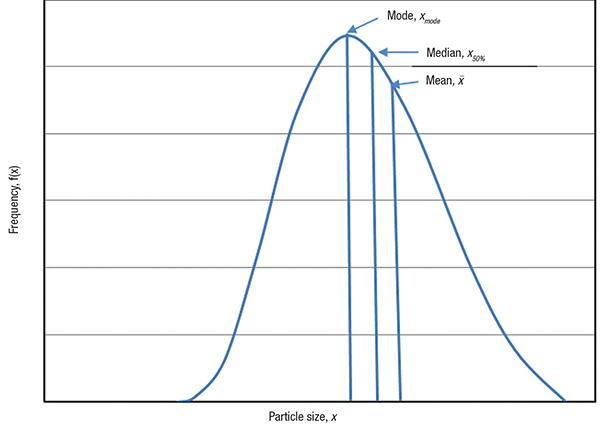
Figure 3. The graph shows representations of mean, median and mode of the particle size distribution
The mean size for the particle population under consideration can be specified in different ways. The common way of defining the mean size is arithmetic mean (Equation 3).
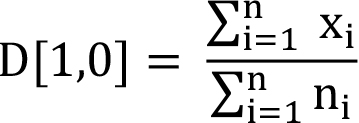 (3)
(3)
Mathematically, it is represented as D[1,0], since the power of the diameter term in the numerator is 1 and that in the denominator is 0 ( xi0 = 1). It is generally advisable to use some kind of weighted mean size to evaluate the effects of particle size on the product quality or performance. For example, weighting of size by surface area is more meaningful for particles where surface area plays significant role (as in the case of catalysts). Equation (4) shows the arithmetic mean weighted by number of particles.
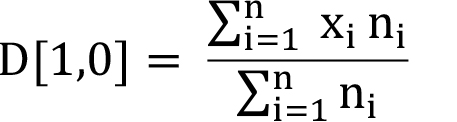 (4)
(4)
It is also possible to calculate mean based on the surface area and volume (Equation (5)):
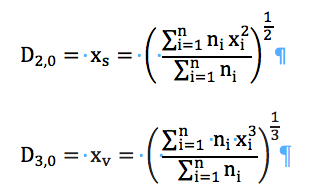 (5)
(5)
The biggest drawback of these formulas is the requirement to calculate a number of particles, which is challenging and difficult task for real applications. However, this issue can be addressed by relating the number to an easily measurable quantity. One probable solution is to relate the number of particles to the mass in the given size interval [3, 4]. The number of particles to the mass in the given size interval can be related as follows:
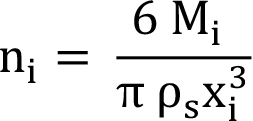 (6)
(6)
Where n i is the number of particles in the i th size class, M i is the mass of the particles in the i th size class, and ρ s is the solid density or particle density. If you substitute Equation (6) into Equation (4), and divide the resulting equation by the total weight of particles ( M t), the final equation is shown in Equation (7), since mass fraction in a given size interval is m i = M i /M t.
 (7)
(7)
Similarly, we can obtain D[2,0] and D[3,0] as follows:
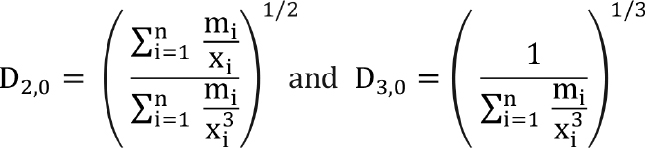 (8)
(8)
The widely used mean size is the surface moment mean (also known as Sauter’s mean).
 (9)
(9)
This can be changed in terms of mass fraction using Equation (6) and Equation (9), as shown in Equation (10):
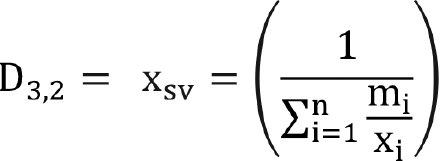 (10)
(10)
The volume moment mean size in Equation (11) is also widely used in chemical engineering. This is also known as DeBroucker’s mean size.
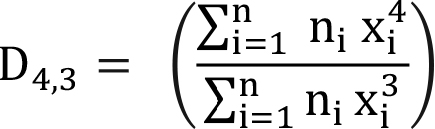 (11)
(11)
Ambiguity can arise when different mean sizes are used. Consider the population of particles containing 10 particles (4 particles of size 1 mm, 3 particles of size 1.5 mm, 3 particles of size 2 mm). We have many choices and can calculate different types of the mean sizes for the same population. It is clear that multiple answers are possible, as shown below for the same population:
D[1,0] = 1.45 mm, D[2,0] = 1.51 mm, D[3,0] = 1.56 mm, D[3,2] = 1.675 mm. All of these are technically correct; the goal should be to select the most suitable value, which is dictated by the end application. For catalyst particles, for example, the equivalent diameter based on the surface-area-to-volume ratio is more appropriate (D[3,2]). For pigment particles, projected area diameter (D[2,0]) may be more relevant. The general definition and other aspects of mean particle size have been described in details in Refs. 1 and 5.
Particle size distributions
Almost all solid materials encountered in real applications involve a range of sizes, so it becomes important to determine particle size distribution (PSD), which is essentially a plot of relative quantity versus particle size. PSD is expressed graphically as a density distribution curve or a cumulative distribution curves. The symbol x is used here to indicate particle size.
In the general method of data representation for PSD, a fraction of the quantity of particles in each size interval is identified. This quantity can be number, length, surface area, volume or mass.
Mathematical representation of particle size. Mathematically, PSD can be described in terms of density distribution or cumulative distribution. In the cumulative distribution, Qr(x) gives the relative amount of particles that are smaller than certain size. If the Qr(x) term can be differentiated, a density distribution qr(x) is obtained. The two are related by the following differential relationship: qr(x) = dQr(x)/dx
The density function, qr (x), is defined such that qr(x)dx represents the fraction of the quantity of particles in a size interval from x to x + dx. Thus, q(x) is the fractional quantity in a size range from x to x + dx, and Qr(x) is a fractional or percentage quantity smaller than size x. The particle size measurement is based on the different measurement principles.
Different size-analysis instruments employ different principles and report different particle characteristics. These characteristics could be number, length, surface area, volume or mass, which depend upon the principles of measurement of the particle size. This gives rise to PSD values that are based on different quantities, including PSD by number, by length, by surface area, and by volume or mass. The PSD by mass and volume are expected to be same if the particle density is independent of size. The integral relation is given in Equation (12):
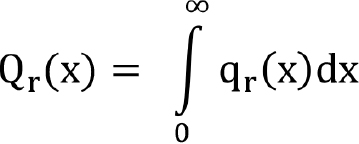 (12)
(12)
Where the subscript r indicates the type of quantity being measured, q represents fractional density and Q represents cumulative density, as per the International Organization for Standardization (ISO) standard 9276 (Representation of Results of Particle Size Analysis). Thus, the following notations are used to indicate different types of PSDs (Table 2).
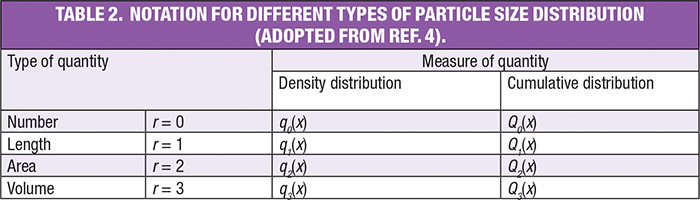
Converting among different PSD
Different particle-size analysis instruments measure different types of PSD. For example, microscopy-based techniques generally give number-based distributions, while laser diffraction measures volume-based distributions, and dynamic light scattering (DLS) measures intensity-based distribution. It is often necessary for a given size distribution based on one type of quantity to be converted into other types for comparisons and other operational purposes. For example, one can convert the number-based distribution into a distribution based on mass or surface area. The following relationships are used to relate different types of distributions [ 1]:
Conversion from number to length: qL (x) = kLqN(x) or q1(x) = kLq0 (x) (13)
Conversion from number to surface area: qS(x) = kSx2 qN(x) or q2 (x) = kSx2 q0(x) (14)
Conversion from number to mass: qM(x) = kM3 qN(x) or q3(x) = kMx3 q0(x) (15)
The values of constants kL, kS and, kM contain information about shape factors, which must be known to make accurate conversions. These factors may vary with particle size and can make it difficult to make accurate conversions. However, the information about the shape factor can be obtained easily, if it is independent of particle size, from the following condition:
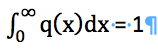 (16)
(16)
The method to determine surface distribution from mass or volume distribution is described below, as in Svarovsky [1] and Rhodes [6].
Converting mass distribution to surface distribution. The method to determine surface distribution from mass distribution is shown below. Using Equations (14) and (15),
qS(x) = kSx2 qN(x) and qM(x) = kMx 3 qN(x)
qS (x) = kS /kM (1/x) qM(x) (17)
and integrating from 0 to x,
![]() (18)
(18)
And from the relation, q(x) = dQ/dx
![]() (19)
(19)
In this way, the surface distribution can be found from Equation (19). The left side of Equation (19) represents the area under the curve (AT) of 1/x versus QM, multiplied by the factor kS /kM. The factor k S /k M, in turn can be found from the normalization condition ∫0 ∞ dQS =1.
Thus, 1 = (kS / kM) AT (20)
Where AT is the area under the curve of 1/ x versus QM). Thus, dividing each value of QM by AT should yield the corresponding values of QS. Similarly, the method to determine mass distribution (QM(x)) from number distribution (QN(x)) is accomplished by multiplying different values of qn, that correspond to different sizes of x, by x3. The resulting curve of x3 qN (x), is then simply scaled down by a following factor to give qM (x) [1].
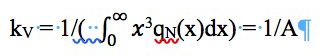 (21)
(21)
Example problem
The example problem in this section is solved to illustrate the procedure of converting between different representations of PSD. The PSD data used in the problem are obtained from a sieve analysis.
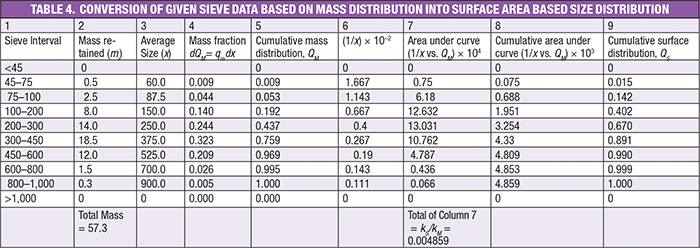
Example Part A. To convert mass distribution into surface-area distribution, tabulate the values of size interval and mass retained in each interval in columns 1 and 2, respectively in Table 3. Determine the average size, as per equation [( x1 + x2) / 2], as shown in column 3.
![]()
The mass fraction is calculated by dividing the mass in each interval by the total mass (57.3 g) and tabulate the result in column 4, using the following equation: mi /(∑ mi).
Then find the cumulative mass fraction by adding values cumulatively and tabulate them in column 5. Find out (1/ x) as in column 6. As explained here, by using Equation (19), we can find the surface distribution:
The integral in the right hand side of the equation represents the area under curve area (AT) of 1/x versus QM, which must be calculated. Here, using the trapezoidal rule, we can find out the required value of the integral in Equation (19).
![]() (22)
(22)
The trapezoidal rule to evaluate the integral is shown below. In our case, we must find out the integral value for each of the size intervals, as well as the total (AT), as shown below. The calculated values have been tabulated in column 7 of Table 4.
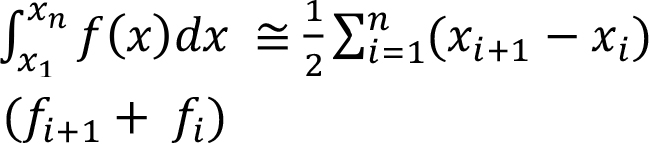 (23)
(23)
For example, the first and second entries in column 7 are
= 0.5(0.009-0) ((1.667+0) x 10–2) = 0.075 × 10–3
= 0.5(0.053-0.009) ((1.143+1.667) x 10-2) = 6.18 x 10–4
The sum of all the entries in the Column 7 gives the value of 1/ kS / kM = 0.004859, as per Equation (20).
Next, calculate the cumulative area under the curve, as shown in Column 8 of Table 4.
Finally, multiply each entry in column 8 by (kS /kM) to yield the surface distribution, as per Equation (19).
0.075 × (1/0.004859) = 0.015, also 0.688 × (1/0.004859) = 0.142
This will result in a cumulative surface distribution, as shown in Column 9.
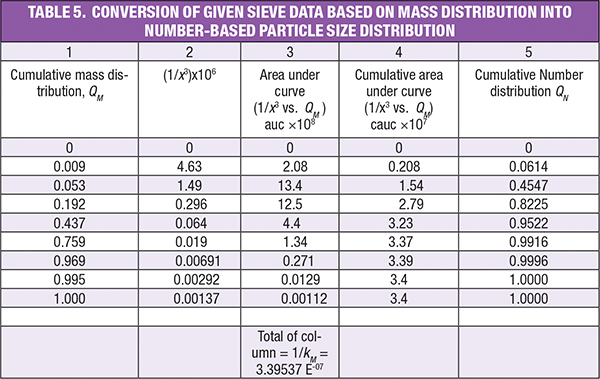
Example Part B. To convert mass distribution into number distribution, a similar procedure is employed, as the two are related by the following relationship, qM (x) = kM x3 qN (x).
Integrating from 0 to x,
∫0x qN (x) dx = 1/ kM ∫0x 1/ x3 qM (x)dx
And from the relation, q(x) = dQ /dx
∫0x dQN = 1/ kM ∫0x 1/x3 dQM
In this way, we can find the number distribution from the mass distribution. The integral can be estimated using the trapezoidal rule, as follows. The remaining steps are similar as illustrated in the case of the solution for Part A. The calculated values can be found in Table 5.
Editor’s note: There is a correction to the formula in the last cell of column 7. It should be “Total of Column 7 = 1/(ks/km) = 0.04859”.
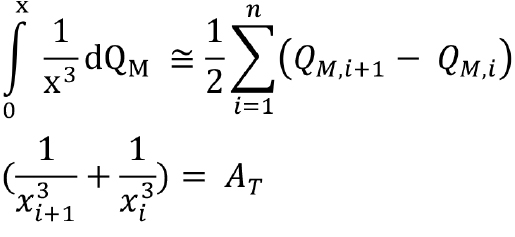
(25)
Example Part C. This example illustrates how to calculate different types of mean sizes. Calculation of mean sizes, such as D[1,0], D[2,0], D[3,0] and D[3,2], are shown for the same dataset. Use columns 3 and 4 of Table 4 for xi and mi, respectively, for the calculations.
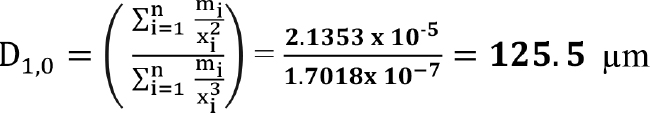 (26)
(26)
Similarly, using Equations (8) and (9), D[2,0] and D[3,0] can be calculated as follows:
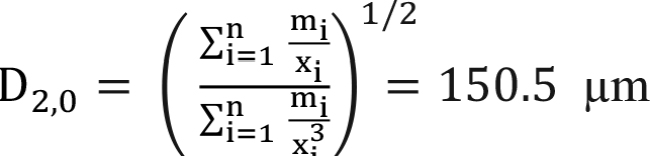 (27)
(27)
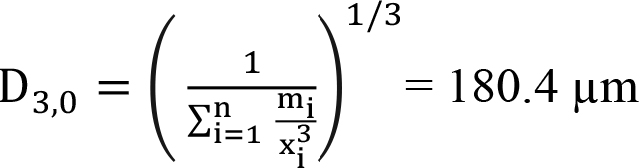 (28)
(28)
Finally, the Sauter mean can also be calculated from Equation (10).
D3,2 = (1/(∑n i=1 (mi /xi)) = 259.4µm (29)
Thus, the same population of particles yield different results depending upon the type of mean required. The plot of the cumulative undersize distribution based on the mass, surface area and number on the same plot is shown in Figure 4. Care must be taken to account for the errors inherent in this type of conversion in the final estimation [5].
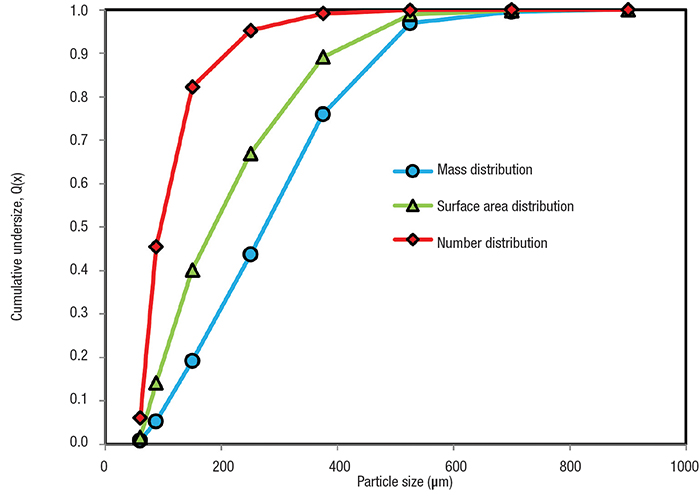
Figure 4. Different ways of measuring particle size distribution (using number, surface area and mass) give rise to different curves for cumulative undersize particles
In most applications in solid-fluid separation, the PSD by mass is of most value, since we are mostly interested in gravimetric efficiencies and mass balances. The PSD by surface or by number may be more relevant in some cases of liquid clarification, where turbidity is an important parameter [1].
When converting from one type of distribution to another, it is suggested that a method be used that gives a suitable PSD to avoid errors inherent in such conversion calculations. The selection of a method should be based on both the particle size and the type of distribution required.
Edited by Scott Jenkins
References:
1. Svarovsky, L. “Solid-Liquid Separation,” 4th ed., Butterworth-Heinemann, Oxford, 2000.
2. Allen, T., “Particle Size Measurement,” Vol. 1, Chapman & Hall, 1997.
3. Sokovnin, O. M., Zagoskina, N. V., Characterizing and Determining Particle Size, Theoretical Foundations of Chemical Engineering, 36, pp. 601–603, 2002.
4. Leschonski, K., Particle size analysis and characterization of a classification process. In “Ullmann’s Encyclopedia of Industrial Chemistry,” edited by Pelc, H., Elvers, B., Hawkins, S., 7th ed., Wiley-VCH, Weinheim, 2005.
5. Rhodes, M. “Introduction to Particle Technology,” 2nd ed., John Wiley & Sons, Chichester, U.K., 2008.
6. Describing the size of the population of particles, Accessed at: http://www.ide.iitkgp.ernet.in/Pedagogy_view/example.jsp?USER_ID=82
Author
 Chetan M. Patel is an assistant professor in the Chemical Engineering Department of S.V. National Institute of Technology (SVNIT; Surat, Gujarat, India; Email: cmp@ched.svnit.ac.in). He obtained a bachelor’s degree in engineering from R.E.C., Surat, a master’s degree from IIT Delhi and a Ph.D. from SVNIT Surat. His research interests include production of nanomaterials by stirred media milling, nanomaterials preparation for drugs, lithium-ion batteries, supercapacitors, powder characterization and handling, discrete element method (DEM) simulations of bulk solids, and molecular simulation of nanocomposites. Patel has 15 years of teaching experience. He developed a pedagogy course on mechanical operations under the National Mission Project and designed many experiments for particulate materials. He delivered many experts talks and has many research papers to his credit in science citation index (SCI/SCIE) journals.
Chetan M. Patel is an assistant professor in the Chemical Engineering Department of S.V. National Institute of Technology (SVNIT; Surat, Gujarat, India; Email: cmp@ched.svnit.ac.in). He obtained a bachelor’s degree in engineering from R.E.C., Surat, a master’s degree from IIT Delhi and a Ph.D. from SVNIT Surat. His research interests include production of nanomaterials by stirred media milling, nanomaterials preparation for drugs, lithium-ion batteries, supercapacitors, powder characterization and handling, discrete element method (DEM) simulations of bulk solids, and molecular simulation of nanocomposites. Patel has 15 years of teaching experience. He developed a pedagogy course on mechanical operations under the National Mission Project and designed many experiments for particulate materials. He delivered many experts talks and has many research papers to his credit in science citation index (SCI/SCIE) journals.