Nuclear magnetic resonance (NMR) spectroscopy is an analytical chemistry technique used to elucidate molecular structure and help identity unknown compounds. It is a powerful research tool used for quality control, reaction monitoring, purity analysis and other functions.
Nuclear spin
The properties of subatomic particles give rise to spin, a form of intrinsic angular momentum that can be represented by imagining the particles as tiny spheres spinning on their axes. Although nuclei don’t actually spin, the concept can help explain the principles underlying NMR.
In atoms where the total number of neutrons or protons is an odd number, the nuclei possess an overall spin. For example, 12C has a spin of 0, while the isotope 13C (6 protons, 7 neutrons), has nonzero spin. NMR spectroscopy only detects nuclei with nonzero spin. Examples include the following: 13C, 1H, 15N, 19F, and 31P.
Magnetic fields
Behaving as spinning charged particles, each NMR-active nuclei generates its own magnetic field, which is oriented in a particular direction. Normally, the magnetic moments generated by a sample are oriented randomly in all possible directions. But in an externally applied magnetic field, the nuclei align their magnetic fields with the applied field — either in the same direction of the applied field (known as the alpha spin state), or aligned against it (beta spin state).
Alpha state is lower energy (more stable and more common) than beta, and the difference between the two is proportional to the strength of the external magnetic field. The strength of an NMR magnet determines spectral resolution. Modern superconducting magnets routinely reach magnetic fields greater than 11 Tesla.
Resonance and relaxation
To generate an NMR spectrum, a sample is placed between the poles of a superconducting magnet and is irradiated with bursts of radio-frequency energy. The radio waves are absorbed by the nuclei when the frequency of the radiation matches the frequency at which the nuclei precess around its axis. When the energy is absorbed, the protons flip from the alpha state into the beta (higher energy) state. When this occurs, the nuclei are said to be in resonance.
When a nuclei returns to its original state from an excited energy state, it is said to undergo “relaxation.” As the nuclei relax, they emit electromagnetic signals whose frequencies can be detected by an NMR spectrometer. The signals (peaks) are plotted on a graph of frequency versus intensity.
Chemical shift
The precise resonant frequency at which the flip from lower to higher energy occurs depends on the effective magnetic field at the nucleus. And the effective magnetic field is influenced by electrons around the nucleus, which shield the nucleus from the applied magnetic field. In general, the more electronegative a nucleus is, the less shielding occurs and the higher the resonant frequency. In this way, the chemical environment surrounding the nucleus, and hence, the chemical structure of a compound, can be probed.
In 1H-NMR, each group of chemically equivalent protons (protons with the same electronic environment) in a molecule gives rise to a separate signal, which appears at a slightly different resonant frequency. These different frequencies are expressed as shifts away from a reference signal, which is set to zero. For 1H-NMR, tetramethylsilane (TMS) is used as the reference signal, and the distance from the reference is called a chemical shift (Figure 1).
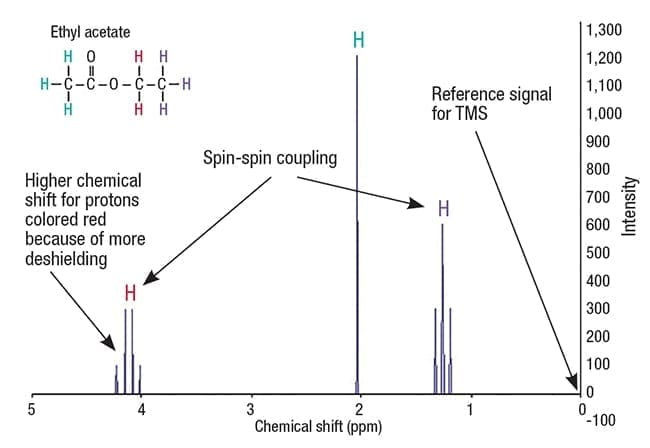
Figure 1. An example H-NMR spectrum for the compound ethyl acetate shows different chemical shifts for the three sets of protons in the compound, each having a different electronic environment
The effective magnetic field experienced by a nucleus also depends on the orientation of neighboring nuclei. This effect, known as spin-spin coupling, results in the splitting of signals into two or more lines. This indicates the number of chemically bonded nuclei near the observed nuclei. Splitting patterns allow experimenters to determine structural information.
Applications
The following are examples of industrial applications for NMR.
Pharmaceuticals. NMR is used for drug discovery, structural confirmation and identifying pharmaceutical effects within natural products
Biofuels. NMR is used to characterize biodiesel, such as quantifying its percentage in fuel mixtures
Polymers. NMR can help observe polymerization reaction completeness and uniformity, qualify relative composition, identify stereoisomers, and determine modular number, molecular weight and polydispersity index
Process analytical technology. NMRs can act as digital detectors for process-line reaction monitoring
Catalysts. NMR spectroscopy can help characterize catalysts deposited on substrates
Petrochemicals. NMR provides information on the identities and characteristics of fluids present in a mixture, and their compositions and viscosities